131.
Study the following reactions and mark the correct properties shown by water.
$$\eqalign{
& \left( {\text{i}} \right)S{O_3} + {H_2}O \to {H_2}S{O_4} \cr
& \left( {{\text{ii}}} \right)C{l_2}{O_7} + {H_2}O \to 2HCl{O_4} \cr
& \left( {{\text{iii}}} \right)CaO + {H_2}O \to Ca{\left( {OH} \right)_2} \cr
& \left( {{\text{iv}}} \right)N{a_2}O + {H_2}O \to 2NaOH \cr} $$
A
All oxides react with water to give hydroxides.
B
Acidic oxides are formed by metals and basic oxides by non-metals.
C
Non-metal oxides combine with water to form acids while metallic oxides combine with water to form alkalies.
D
Acidic oxides are stronger than basic oxides since they form strong acids.
Answer :
Non-metal oxides combine with water to form acids while metallic oxides combine with water to form alkalies.
132. Which of the following is an atom of tritium?
A


B
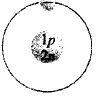

C
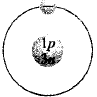

D


Answer :


133. When a substance $$A$$ reacts with water it produces a combustible gas $$B$$ and a solution of substance $$C$$ in water. When another substance $$D$$ reacts with this solution of $$C,$$ it also produces the same gas $$B$$ on warming but $$D$$ can produce gas $$B$$ on reaction with dilute sulphuric acid at room temperature. $$A$$ imparts a deep golden yellow colour to a smokeless flame of Bunsen burner. $$A, B, C$$ and $$D$$ respectively are
A
$$Na,{H_2},NaOH,Zn$$
B
$$K,{H_2},KOH,Al$$
C
$$Ca,{H_2},Ca{\left( {OH} \right)_2},Sn$$
D
$$Ca{C_2},{C_2}{H_2},Ca{\left( {OH} \right)_2},Fe$$
Answer :
$$Na,{H_2},NaOH,Zn$$
134. The amount of $${H_2}{O_2}$$ present in 1 litre of $$1.5\,N\,{H_2}{O_2}$$ solution, is :
A
25.5$$\,g$$
B
3.0$$\,g$$
C
8.0$$\,g$$
D
2.5$$\,g$$
Answer :
25.5$$\,g$$
135. Radioactive elements emit $$\alpha ,\beta $$ and $$\gamma $$ rays and are characterised by their half - lives. The radioactive isotope of hydrogen is
A
protium
B
deuterium
C
tritium
D
hydronium
Answer :
tritium
136. Which of the following compounds is used for water softening?
A
$$C{a_3}{\left( {P{O_4}} \right)_2}$$
B
$$N{a_3}P{O_4}$$
C
$$N{a_6}{P_6}{O_{18}}$$
D
$$N{a_2}HP{O_4}$$
Answer :
$$N{a_6}{P_6}{O_{18}}$$
137. Which of the following is wrong about $${H_2}{O_2}?$$ It is used
A
as an aerating agent in production of sponge rubber
B
as an antichlor
C
for restoring white colour of blackened lead painting
D
None of these
Answer :
None of these
138. In a permutit, the calcium and magnesium ions of hard water are exchanged by
A
$$CO_3^{2 - }$$ and $$HCO_3^ - $$ ions of permutit
B
$$N{a^ + }$$ ions of permutit
C
$$A{l^{3 + }}$$ ions of permutit
D
$$S{i^{4 + }}$$ ions of permutit
Answer :
$$N{a^ + }$$ ions of permutit
139. Which compound is formed when calcium carbide reacts with heavy water?
A
$${C_2}{D_2}$$
B
$$Ca{D_2}$$
C
$$C{D_2}$$
D
$$C{a_2}{D_2}$$
Answer :
$${C_2}{D_2}$$
140. In context with the industrial preparation of hydrogen from water gas $$\left( {CO + {H_2}} \right),$$ which of the following is the correct statement ?
A
$$CO$$ and $${H_2},$$ are fractionally separated using differences in their densities
B
$$CO$$ is removed by absorption in aqueous $$C{u_2}C{l_2}$$ solution
C
$${H_2}$$ is removed through occlusion with $$Pd$$
D
$$CO$$ is oxidised to $$C{O_2}$$ with steam in the presence of a catalyst followed by absorption of $$C{O_2}$$ in alkali
Answer :
$$CO$$ is oxidised to $$C{O_2}$$ with steam in the presence of a catalyst followed by absorption of $$C{O_2}$$ in alkali