151. $$A,$$ 6 volume sample of $${H_2}{O_2}$$
A
will contain $$6\% \,\,V/V$$ of $${H_2}{O_2}$$
B
will contain $$6\% \,\,W/V$$ of $${H_2}{O_2}$$
C
would give 6 volumes of oxygen per unit volume of $${H_2}{O_2}$$ sample at $$STP$$
D
would give 6 volumes of oxygen per unit weight of $${H_2}{O_2}$$ sample at $$STP$$
Answer :
would give 6 volumes of oxygen per unit volume of $${H_2}{O_2}$$ sample at $$STP$$
152.
Which of the statements given below are true for the structure of water molecule?
(i) Oxygen undergoes $$s{p^3}$$ hybridisation.
(ii) Due to presence of two lone pairs of electrons on oxygen the $$H - O - H$$ bond angle is $${118.4^ \circ }.$$
(iii) Due to angular geometry the net dipole moment of water is not zero, $$\mu = 1.84\,D.$$
A
(i) and (ii)
B
(ii) and (iii)
C
(i) and (iii)
D
Only (ii)
Answer :
(i) and (iii)
153.
Consider the reactions :
$$\left( {\text{i}} \right){H_2}{O_2} + 2HI \to {I_2} + 2{H_2}O$$
$$\left( {{\text{ii}}} \right)HOCl + {H_2}{O_2} \to $$ $${H_3}{O^ + } + C{l^ - } + {O_2}$$
Which of the following statements is correct about $${H_2}{O_2}$$ with reference to these reactions? Hydrogen peroxide is ___________.
A
an oxidising agent in both (i) and (ii)
B
an oxidising agent in (i) and reducing agent in (ii)
C
a reducing agent in (i) and oxidising agent in (ii)
D
a reducing agent in both (i) and (ii)
Answer :
an oxidising agent in (i) and reducing agent in (ii)
154. Which of the following is not a property of hydrogen?
A
It is a colourless, odourless gas.
B
It is highly combustible.
C
It is highly poisonous gas.
D
It is lighter than air.
Answer :
It is highly poisonous gas.
155. The order of reactivity of halogens towards hydrogen is
A
$${F_2} > C{l_2} > B{r_2} > {I_2}$$
B
$${I_2} > B{r_2} > C{l_2} > {F_2}$$
C
$$C{l_2} > B{r_2} > {I_2} > {F_2}$$
D
$$B{r_2} > C{l_2} > {F_2} > {I_2}$$
Answer :
$${F_2} > C{l_2} > B{r_2} > {I_2}$$
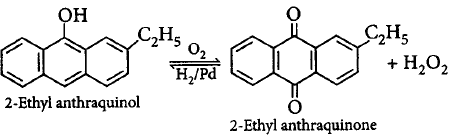
156. Which of the following reagents can be used for the preparation of hydrogen peroxide
A
Sodium peroxide
B
2 - Ethylanthraquinol
C
Barium peroxide
D
All of these
Answer :
All of these
157. A water sample is said to contain permanent hardness if water contains
A
sulphates and chlorides of calcium and magnesium
B
carbonates of calcium and magnesium
C
bicarbonates of calcium and magnesium
D
sulphates and chlorides of sodium and potassium
Answer :
sulphates and chlorides of calcium and magnesium
158. $$5.0\,c{m^3}$$ of $${H_2}{O_2}$$ liberates $$0.508\,g$$ of iodine from an acidified $$KI$$ solution. The strength of $${H_2}{O_2}$$ solution in terms of volume strength at $$STP$$ is
A
6.48 volumes
B
4.48 volumes
C
7.68 volumes
D
none of these
Answer :
4.48 volumes
159. The boiling point of heavy water is
A
$$100{\,^ \circ }C$$
B
$$101.4{\,^ \circ }C$$
C
$$99{\,^ \circ }C$$
D
$$110{\,^ \circ }C$$
Answer :
$$101.4{\,^ \circ }C$$
160.
Match the reactions of column I with their types given in column II and mark the appropriate choice.
| Column I | Column II | ||
|---|---|---|---|
| a. | $${H_2}O + N{H_3} \rightleftharpoons NH_4^ + + O{H^ - }$$ | 1. | Self ionisation of $${H_2}O$$ |
| b. | $$FeC{l_3} + 3{H_2}O \to Fe{\left( {OH} \right)_3} + 3HCl$$ | 2. | Decomposition |
| c. | $${H_2}O + {H_2}O \rightleftharpoons {H_3}{O^ + } + O{H^ - }$$ | 3. | Acidic nature of $${H_2}O$$ |
| d. | $$2{H_2}O \to 2{H_2} + {O_2}$$ | 4. | Hydrolysis |
A
a - 2, b - 1, c - 3, d - 4
B
a - 3, b - 2, c - 4, d - 1
C
a - 1, b - 2, c - 4, d - 3
D
a - 3, b - 4, c - 1, d - 2
Answer :
a - 3, b - 4, c - 1, d - 2