11.
An alkali is titrated against an acid with methyl orange as indicator, which of the following in a correct combination?

A
(a)
B
(b)
C
(c)
D
(d)
Answer :
(c)
12. In Lassaigne’s test, the organic compound is fused with a piece of sodium metal in order to
A
increase the ionisation of the compound.
B
decrease the melting point of the compound.
C
increase the reactivity of the compound.
D
convert the covalent compound into a mixture of ionic compounds.
Answer :
convert the covalent compound into a mixture of ionic compounds.
13. Lassaigne’s test is used to detect
A
nitrogen
B
sulphur
C
chlorine
D
All of the above
Answer :
All of the above
14. An organic compound has $$C$$ and $$H$$ percentage in the ratio 6 : 1 and $$C$$ and $$O$$ percentage in the ratio 3 : 4 the compound is
A
$$HCHO$$
B
$$C{H_3}OH$$
C
$$C{H_3}C{H_2}OH$$
D
$${\left( {COOH} \right)_2}$$
Answer :
$$HCHO$$
15. Lassaigne’s test for the detection of nitrogen fails in
A
$$N{H_2}CONHN{H_2} \cdot HCl$$
B
$$N{H_2}N{H_2} \cdot HCl$$
C
$$N{H_2}CON{H_2}$$
D
$${C_6}{H_5}NHN{H_2} \cdot HCl$$
Answer :
$$N{H_2}N{H_2} \cdot HCl$$
16. $$A$$ is a lighter phenol and $$B$$ is an aromatic carboxylic acid. Separation of a mixture of $$A$$ and $$B$$ can be carried out easily by using a solution of
A
sodium hydroxide
B
sodium sulphate
C
calcium chloride
D
sodium bicarbonate
Answer :
sodium bicarbonate
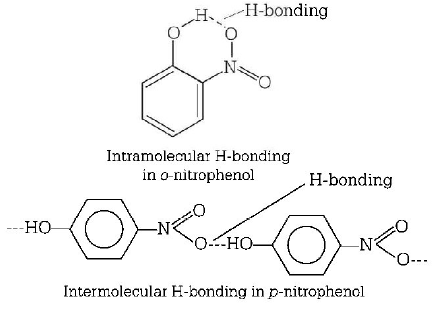
17. The most suitable method of separation of 1 : 1 mixture of $$ortho$$ and $$para$$ - nitrophenols is
A
sublimation
B
chromatography
C
crystallisation
D
steam distillation
Answer :
steam distillation
18. $$A$$ is a lighter phenol and $$B$$ is an aromatic carboxylic acid. Separation of mixture of $$A$$ and $$B$$ can be carried out easily by using a solution of
A
sodium hydroxide
B
sodium sulphate
C
calcium chloride
D
sodium bicarbonate
Answer :
sodium bicarbonate
19. The Lassaigne’s extract is boiled with $$conc.\,HN{O_3}$$ while testing for halogens. By doing so it
A
helps in the precipitation of $$AgCl$$
B
increases the solubility product of $$AgCl$$
C
increases the concentration of $$NO_3^ - \,ions$$
D
decomposes $$N{a_2}S$$ and $$NaCN,$$ if formed
Answer :
decomposes $$N{a_2}S$$ and $$NaCN,$$ if formed
20. $$1.25\,g$$ of a sample of $$N{a_2}C{O_3}$$ and $$N{a_2}S{O_4}$$ is dissolved in $$250\,ml$$ solution. $$25\,ml$$ of this solution neutralises $$20\,ml$$ of $$0.1\,N\,{H_2}S{O_4}.$$ The $$\% $$ of $$N{a_2}C{O_3}$$ in this sample is
A
84.8%
B
8.48%
C
15.2%
D
42.4%
Answer :
84.8%