21.
$$KMn{O_4}$$ reacts with oxalic acid as : $$MnO_4^ - + {C_2}O_4^{2 - } + {H^ + } \to $$ $$M{n^{2 + }} + C{O_2} + {H_2}O$$
Hence, $$50\,ml$$ of $$0.04\,M\,KMn{O_4}$$ is acidic medium is chemically equivalent to
A
$$100\,ml\,\,{\text{of}}\,\,0.1\,M\,{H_2}{C_2}{O_4}$$
B
$$50\,ml\,\,{\text{of}}\,\,0.2\,M\,{H_2}{C_2}{O_4}$$
C
$$50\,ml\,\,{\text{of}}\,\,0.1\,M\,{H_2}{C_2}{O_4}$$
D
$$25\,ml\,\,{\text{of}}\,\,0.1\,M\,{H_2}{C_2}{O_4}$$
Answer :
$$50\,ml\,\,{\text{of}}\,\,0.1\,M\,{H_2}{C_2}{O_4}$$
22. In Kjeldahl’s method, $$CuS{O_4}$$ acts as
A
oxidising agent
B
reducing agent
C
hydrolysing agent
D
catalytic agent
Answer :
catalytic agent
23. Copper wire test for halogens is known as
A
Duma’s Test
B
Beilstein’s Test
C
Liebig’s Test
D
Lassigne’s Test
Answer :
Beilstein’s Test
24. In sodium fusion test of organic compounds, the nitrogen of the organic compound is converted into
A
sodamide
B
sodium cyanide
C
sodium nitrite
D
sodium nitrate
Answer :
sodium cyanide
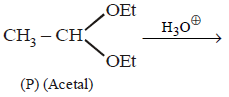
25.
Compound 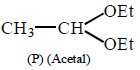
and 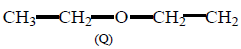 can be differentiated by :
can be differentiated by :
A
$${H_3}{O^ \oplus },Na$$
B
$${H_3}{O^ \oplus },$$ Tollens' test
C
$${H_3}{O^ \oplus },$$ Fehling test
D
All of these
Answer :
All of these
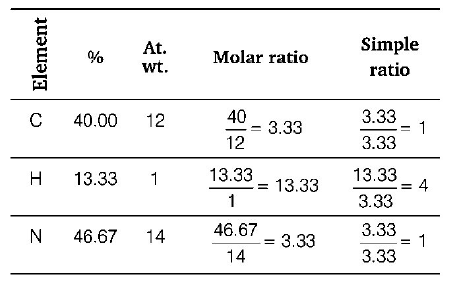
26. An organic compound containing $$C, H$$ and $$N$$ gave the following results on analysis $$C = 40\% ,H = 13.33\% ,N = 46.67\% .$$ Its empirical formula would be
A
$${C_2}{H_7}{N_2}$$
B
$$C{H_5}N$$
C
$$C{H_4}N$$
D
$${C_2}{H_7}N$$
Answer :
$$C{H_4}N$$
27. $$0.45\,g$$ of acid ( molecular weight 90 ) is neutralised by $$20\,ml$$ of $$0.5\,N$$ caustic potash. The basicity of acid is
A
1
B
2
C
3
D
4
Answer :
2
28. Aniline is usually purified by
A
chromatographic technique
B
steam distillation
C
by addition of oxalic acid
D
fractional crystallization
Answer :
steam distillation
29. The formula of gas is $${\left[ {CO} \right]_x}.$$ If its vapour density is 140, the value of $$x$$ will be :
A
2.5
B
3.0
C
5.0
D
6.0
Answer :
5.0
30. The best method for the separation of naphthalene and benzoic acid from their mixture is :
A
Distillation
B
Sublimation
C
Chromatography
D
Crystallisation
Answer :
Sublimation