31.
In Kjeldahl’s method for the estimation of $${N_2},$$ potassium sulphate and copper sulphate are used. On the basis of their functions which of the following statement $$(s)$$ is/are correct ?
(i) Potassium sulphate raises the $$bpt.$$ and ensures complete reaction.
(ii) Copper sulphate acts as catalyst.
(iii) Potassium sulphate acts as catalyst and copper sulphate raises the $$bpt.$$
A
Only (iii) is correct
B
(i) and (ii) are correct
C
Only (ii) is correct
D
None is correct
Answer :
(i) and (ii) are correct
32. The compound formed in the positive test for nitrogen with the Lassaigne solution of an organic compound is
A
$$F{e_4}{\left[ {Fe{{\left( {CN} \right)}_6}} \right]_3}$$
B
$$N{a_3}\left[ {Fe{{\left( {CN} \right)}_6}} \right]$$
C
$$Fe{\left( {CN} \right)_3}$$
D
$$N{a_4}\left[ {Fe{{\left( {CN} \right)}_5}NOS} \right]$$
Answer :
$$F{e_4}{\left[ {Fe{{\left( {CN} \right)}_6}} \right]_3}$$
33. Volume of $$3\% $$ solution of sodium carbonate necessary to neutralise a litre of $$0.1\,N$$ sulphuric acid
A
176.66$$\,ml$$
B
156.6$$\,ml$$
C
116.0$$\,ml$$
D
196.1$$\,ml$$
Answer :
176.66$$\,ml$$
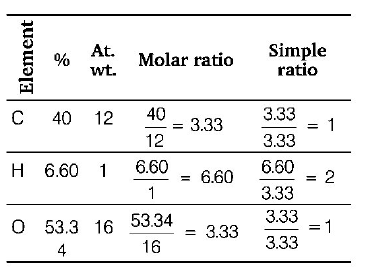
34. An organic compound contains $$C = 40\% ,O = 53.34\% $$ and $$H = 6.60\% .$$ The empirical formula of the compound is
A
$$C{H_2}O$$
B
$$CHO$$
C
$$C{H_4}{O_2}$$
D
$${C_2}{H_2}O$$
Answer :
$$C{H_2}O$$
35. In steam distillation of toluene, the pressure of toluene in vapour is
A
equal to the pressure of barometer
B
less than the pressure of barometer
C
equal to vapour pressure of toluene in simple distillation
D
more than vapour pressure of toluene in simple distillation
Answer :
less than the pressure of barometer
36. If $$0.2\,$$ $$gram$$ of an organic compound containing carbon, hydrogen and oxygen on combustion, yielded $$0.147$$ $$gram$$ carbon dioxide and $$0.12$$ $$gram$$ water. What will be the content of oxygen in the substance ?
A
73.29%
B
78.45%
C
83.23%
D
89.50%
Answer :
73.29%
37. $$0.59\,g$$ of an organic substance when treated with caustic soda evolved ammonia which required $$20\,cc$$ of $$N/2$$ sulphuric acid for neutralisation. The percentage of nitrogen is
A
26.32$$\% $$
B
40$$\% $$
C
53.6$$\% $$
D
63.6$$\% $$
Answer :
26.32$$\% $$
38. Camphor is often used in molecular mass determination because
A
it is readily available
B
it has a very high cryoscopic constant
C
it is volatile
D
it is solvent for organic substances
Answer :
it is volatile
39. $$0.75\,g$$ platinic chloride of a mono- acidic base on ignition gives $$0.245\,g$$ platinum. The molecular weight of the base is
A
75.0
B
93.5
C
100
D
80.0
Answer :
93.5
40. Kjeldahl’s method is used in the estimation of
A
nitrogen
B
halogens
C
sulphur
D
oxygen
Answer :
nitrogen