261. Tertiary butyl alcohol gives tertiary butyl chloride on treatment with
A
$$\frac{{{\text{conc}}{\text{.}}\,HCl}}{{{\text{anhydrous }}\,ZnC{l_2}}}$$
B
$$KCN$$
C
$$NaOCl$$
D
$$C{l_2}$$
Answer :
$$\frac{{{\text{conc}}{\text{.}}\,HCl}}{{{\text{anhydrous }}\,ZnC{l_2}}}$$
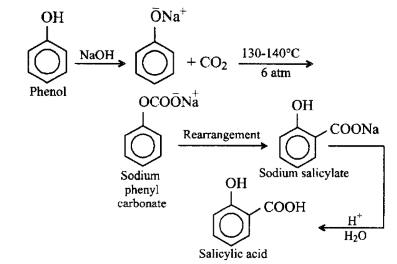
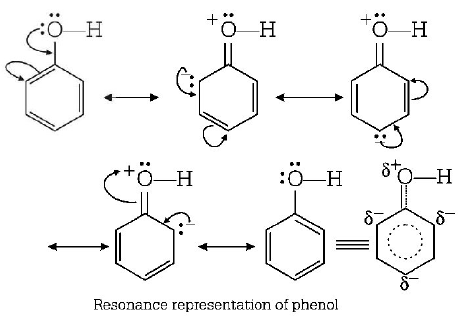
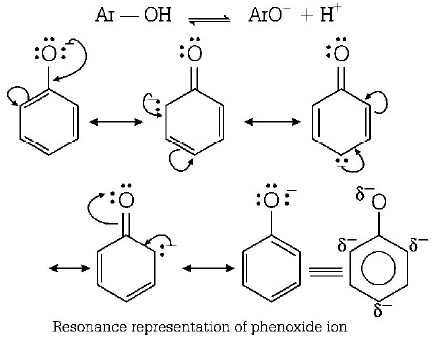
262. The major product obtained on interaction of phenol with sodium hydroxide and carbon dioxide is
A
benzoic acid
B
salicylaldehyde
C
salicylic acid
D
phthalic acid
Answer :
salicylic acid
263.
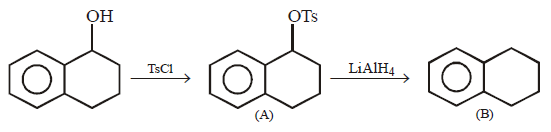
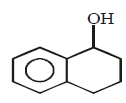 \[\xrightarrow[\text{Pyridine}]{TsCl}\left( A \right)\xrightarrow{LiAl{{H}_{4}}}\left( B \right)\]
\[\xrightarrow[\text{Pyridine}]{TsCl}\left( A \right)\xrightarrow{LiAl{{H}_{4}}}\left( B \right)\]
Product $$(B)$$ of the above reaction is :
A
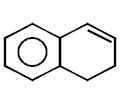

B
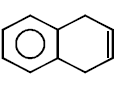

C
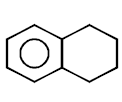

D
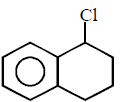

Answer :


264.
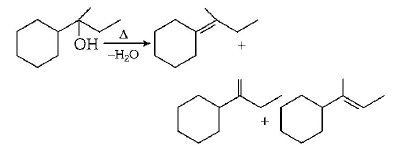
Which of the following is not the product of dehydration of 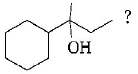
A
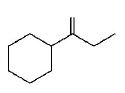

B
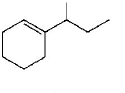

C
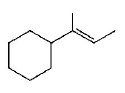

D
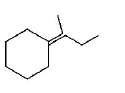

Answer :


265. Which of the following species can act as the strongest base?
A
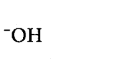
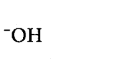
B
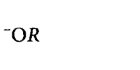
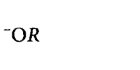
C
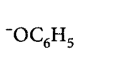
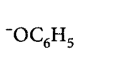
D
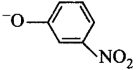

Answer :
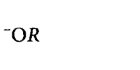
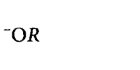
266. A compound $${C_6}{H_{14}}{O_2}$$ has two tertiary alcoholic groups. The IUPAC name of this compound is
A
2, 3-dimethyl - 1, 2-butanediol
B
3, 3-dimethyl - 1, 2-butanediol
C
2, 3-dimethyl - 2, 3-butanediol
D
2-methyl - 2, 3-pentanediol
Answer :
2, 3-dimethyl - 2, 3-butanediol
267.
The correct acidic order of following is
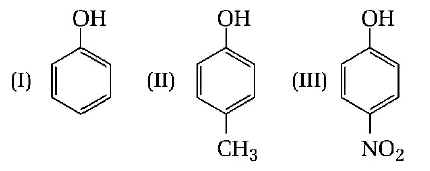
A
I > II > III
B
III > I > II
C
II > III > I
D
I > III > II
Answer :
III > I > II
268. A compound that gives a positive iodoform test is
A
1 - pentanol
B
2 - pentanone
C
3 - pentanone
D
pentanal
Answer :
2 - pentanone
269. Propene, $$C{H_3} - CH = C{H_2}$$ can be converted into 1-propanol by oxidation. Indicate which set of reagents amongst the following is ideal to affect the above conversion?
A
$$KMn{O_4}$$ (alkaline)
B
Osmium tetroxide $$\left( {\frac{{Os{O_4}}}{{C{H_2}C{l_2}}}} \right)$$
C
$${B_2}{H_6}$$ and $$alk\,{H_2}{O_2}$$
D
$$\frac{{{O_3}}}{{Zn}}$$
Answer :
$${B_2}{H_6}$$ and $$alk\,{H_2}{O_2}$$
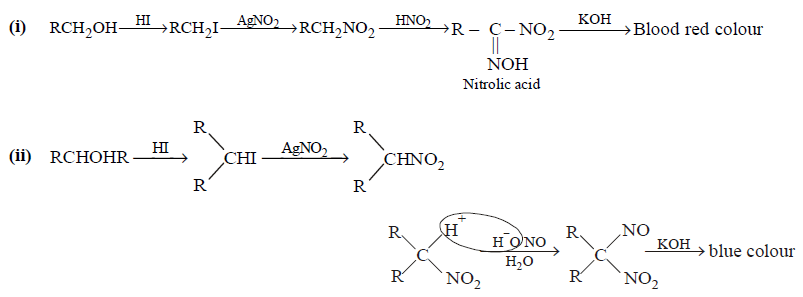
270. In the Victor-Meyer’s test, the colour given by $${1^ \circ },{2^ \circ }$$ and $${3^ \circ }$$ alcohols are respectively :
A
Red, colourless, blue
B
Red, blue, colourless
C
Colourless, red, blue
D
Red, blue, violet
Answer :
Red, blue, colourless