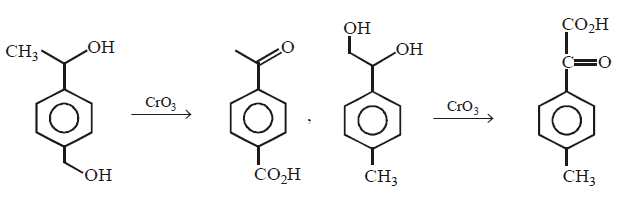
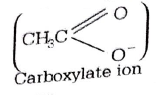
271.
An optically active alcohol of formula $${C_9}{H_{12}}{O_2}$$ produced the following compound when refluxed with $$KMn{O_4}.$$
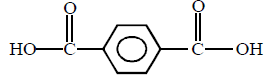
The original compound showed these properties also :
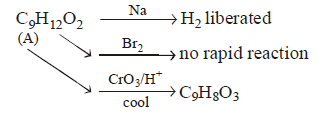
What is structure of $$(A)?$$
A
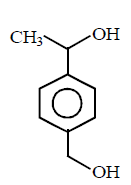

B
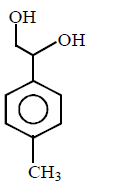

C
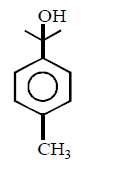

D
Answer :
272.
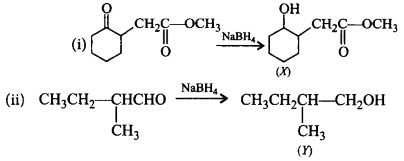
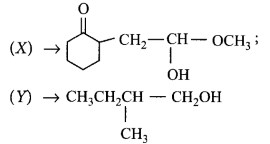
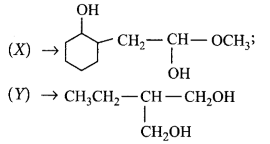
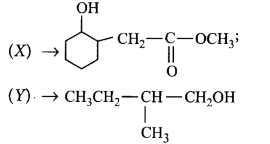
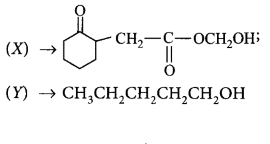
Choose the correct $$X$$ and $$Y$$ in the given reactions.
$$\left( {\text{i}} \right)$$ 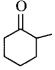 \[C{{H}_{2}}\underset{\begin{smallmatrix}
\parallel \\
O
\end{smallmatrix}}{\mathop{-C-}}\,OC{{H}_{3}}\xrightarrow{NaB{{H}_{4}}}\left( X \right)\]
\[C{{H}_{2}}\underset{\begin{smallmatrix}
\parallel \\
O
\end{smallmatrix}}{\mathop{-C-}}\,OC{{H}_{3}}\xrightarrow{NaB{{H}_{4}}}\left( X \right)\]
\[\left( \text{ii} \right)\underset{\begin{smallmatrix}
|\,\,\, \\
\,C{{H}_{3}}
\end{smallmatrix}}{\mathop{C{{H}_{3}}C{{H}_{2}}CH-CHO}}\,\] \[\xrightarrow{NaB{{H}_{4}}}\left( Y \right)\]
A

B

C

D

Answer :


273.
In the following reaction sequence,
\[C{{H}_{3}}\underset{\begin{smallmatrix}
|\,\,\,\,\,\, \\
OH\,\,\,
\\
\left( X \right)
\end{smallmatrix}}{\mathop{-CH-}}\,C{{H}_{3}}\xrightarrow{\left[ O \right]}Y\] \[\xrightarrow[\frac{{{H}^{+}}}{{{H}_{2}}O}]{C{{H}_{3}}MgBr}Z;Z\] is
A
butan-1-ol
B
butan-2-ol
C
2-methylpropan-2-ol
D
1, 1-dimethylethanol
Answer :
2-methylpropan-2-ol
274. The best method to prepare 3-methylbutan-2-ol from 3-methylbut-1-ene is
A
addition of water in presence of dil. $${H_2}S{O_4}$$
B
addition of $$HCl$$ followed by reaction with dil. $$NaOH$$
C
hydroboration - oxidation reaction
D
Reimer-Tiemann reaction
Answer :
addition of water in presence of dil. $${H_2}S{O_4}$$
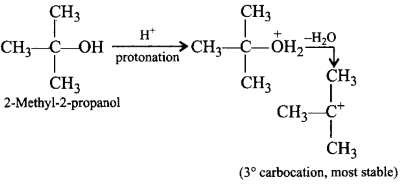
275. Which of the following alcohols will give the most stable carbocation during dehydration?
A
2-Methyl-1-propanol
B
2-Methyl-2-propanol
C
1-Butanol
D
2-Butanol
Answer :
2-Methyl-2-propanol
276. Amongst the following alcohols which would react fastest with $$conc.\,HCl$$ and $$ZnC{l_2}?$$
A
pentanol
B
2 - methyl butanol
C
2 - pentanol
D
2 - methyl butan - 2 - $$ol$$
Answer :
2 - methyl butan - 2 - $$ol$$
277.
Match the column I with column II and mark the appropriate choice.
| Column I | Column II | ||
|---|---|---|---|
| a. | Methanol | 1. | Conversion of phenol to $$o$$ - hydroxysalicylic acid |
| b. | Kolbe's reaction | 2. | Ethyl alcohol |
| c. | Williamson's synthesis | 3. | Conversion of phenol to salicylaldehyde |
| d. | Conversion of 1° alcohol to aldehyde | 4. | Wood spirit |
| e. | Reimer-Tiemann reaction | 4. | Heated copper at 573 $$K$$ |
| f. | Fermentation | 4. | Reaction of alkyl halide with sodium alkoxide |
A
a - 2, b - 4, c - 1, d - 3, e - 5, f - 6
B
a - 6, b - 5, c - 2, d - 4, e - 1, f - 3
C
a - 4, b - 1, c - 6, d - 5, e - 3, f - 2
D
a - 5, b - 2, c - 4, d - 6, e - 3, f - 1
Answer :
a - 4, b - 1, c - 6, d - 5, e - 3, f - 2
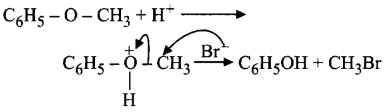
278.
In the reaction, 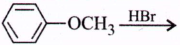 the products are
the products are
A
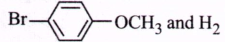
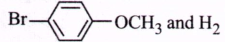
B


C


D


Answer :


279.
Match the column I with column II and mark the appropriate choice.
| Column I | Column II | ||
|---|---|---|---|
| a. | Hydrolysis of benzene diazonium chloride | 1. | $$p$$ - Cresol |
| b. | Phenol + methyl chloride in presence of anh. $$AlC{l_3}$$ | 2. | Salicylic acid |
| c. | Reaction of sodium phenoxide with $$C{O_2}$$ | 3. | Picric acid |
| d. | Phenol + Conc. $$HN{O_3}$$ | 4. | Phenol |
A
a - 1, b - 3, c - 2, d - 4
B
a - 2, b - 3, c - 4, d - 1
C
a - 4, b - 1, c - 2, d - 3
D
a - 3, b - 4, c - 1, d - 2
Answer :
a - 4, b - 1, c - 2, d - 3
280. Given are cyclohexanol (i), acetic acid (ii), 2, 4, 6 - trinitrophenol (iii) and phenol (iv). In these, the order of decreasing acidic character will be
A
(iii) > (ii) > (iv) > (i)
B
(ii) > (iii) > (i) > (iv)
C
(ii) > (iii) > (iv) > (i)
D
(iii) > (iv) > (ii) > (i)
Answer :
(iii) > (ii) > (iv) > (i)