151. Which of the following can possibly be used as an analgesic without causing addiction?
A
Morphine
B
$$N$$ - acetyl - $$para$$ - aminophenol
C
Diazepam
D
None of these
Answer :
$$N$$ - acetyl - $$para$$ - aminophenol
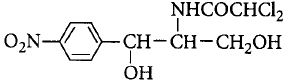
152.
The structure given below is known as
A
prontosil
B
sulphapyridine
C
chloramphenicol
D
chloroxylenol
Answer :
chloramphenicol
153. Soaps do not work in hard water containing calcium and magnesium ions because
A
$$C{a^{2 + }}$$ and $$M{g^{2 + }}$$ ions form insoluble calcium and magnesium salts in the form of scum
B
$$N{a^ + }$$ and $${K^ + }$$ present in soap react with $$C{a^{2 + }}$$ and $$M{g^{2 + }}$$ and hinder cleansing process
C
a large amount of soap is to be used in presence of $$C{a^{2 + }}$$ and $$M{g^{2 + }}$$
D
scum formed by combination of $$N{a^ + },C{a^{2 + }}$$ and $$M{g^{2 + }}$$ stick to the cloth and are not removed on agitation
Answer :
$$C{a^{2 + }}$$ and $$M{g^{2 + }}$$ ions form insoluble calcium and magnesium salts in the form of scum
154. Which of the following statements about aspirin is not true ?
A
It is effective in relieving pain.
B
It is a neurologically active drug.
C
It has antiblood clotting action.
D
It belongs to narcotic analgesics.
Answer :
It belongs to narcotic analgesics.