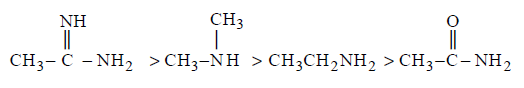121. A compound $$'A'$$ when treated with \[HN{{O}_{3}}\] ( in presence of \[{{H}_{2}}S{{O}_{4}}\] ) gives compound $$'B',$$ which is then reduced with $$Sn$$ and $$HCl$$ to aniline ? The compound $$'A'$$ is
A
toluene
B
benzene
C
ethane
D
acetamide
Answer :
benzene
122. The source of nitrogen in Gabriel synthesis of amines is
A
sodium azide, $$Na{N_3}$$
B
sodium nitrite, $$NaN{O_2}$$
C
potassium cyanide, $$KCN$$
D
potassium phthalimide, $${C_6}{H_4}{\left( {CO} \right)_2}{N^ - }{K^ + }$$
Answer :
potassium phthalimide, $${C_6}{H_4}{\left( {CO} \right)_2}{N^ - }{K^ + }$$
123. Tertiary amines have lowest boiling points amongst isomeric amines because
A
they have highest molecular mass
B
they do not form hydrogen bonds
C
they are more polar in nature
D
they are most basic in nature
Answer :
they do not form hydrogen bonds
124. Phenyl isocyanides are prepared from which of the following reaction?
A
Rosenmund's reaction
B
Carbylamine reaction
C
Reimer - Tiemann reaction
D
Wurtz reaction
Answer :
Carbylamine reaction
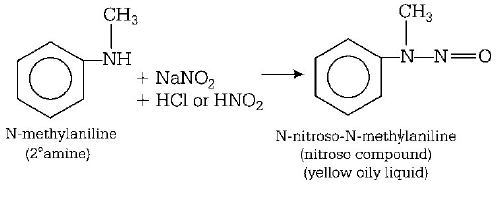
125. The compound which on reaction with aqueous nitrous acid at low temperature produces an oily nitrosamine is
A
methylamine
B
ethylamine
C
diethylamine
D
triethylamine
Answer :
diethylamine
126.
Predict the product
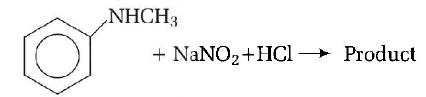
A
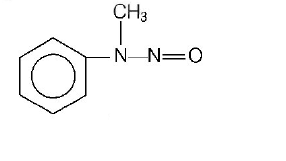

B
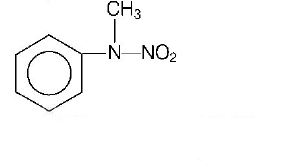

C
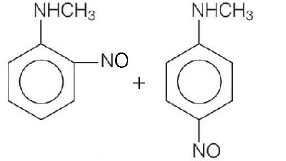

D
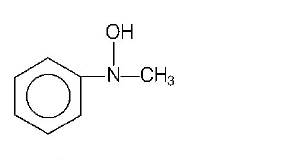

Answer :


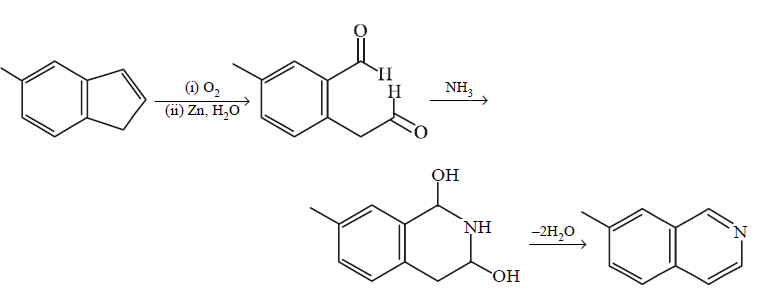
127.
In the following reactions, the product $$S$$ is
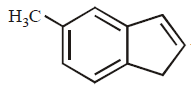 \[\xrightarrow[\text{ii}.\,Zn,\,{{H}_{2}}O]{\text{i}.\,{{O}_{3}}}R\xrightarrow{N{{H}_{3}}}S\]
\[\xrightarrow[\text{ii}.\,Zn,\,{{H}_{2}}O]{\text{i}.\,{{O}_{3}}}R\xrightarrow{N{{H}_{3}}}S\]
A
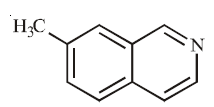

B
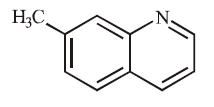

C
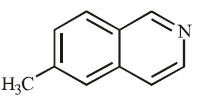

D
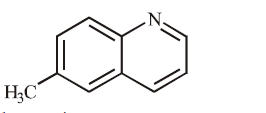

Answer :


128. When aniline is heated with conc. $${H_2}S{O_4}$$ at $$455 - 475\,K,$$ it forms
A
aniline hydrogensulphate
B
sulphanilic acid
C
amino benzene sulphonic acid
D
benzenesulphonic acid
Answer :
sulphanilic acid
129.
Identify the correct pathway to convert propanoic acid to ethylamine. The reagent represented by $$A, B$$ and $$C$$ are \[C{{H}_{3}}C{{H}_{2}}COOH\xrightarrow{A}X\xrightarrow{B}\] \[Y\xrightarrow{C}C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
| $$A$$ | $$B$$ | $$C$$ | |
|---|---|---|---|
| (a) | $$Ca{\left( {OH} \right)_2}$$ | Heat | $$Pt/{H_2}$$ |
| (b) | $$SOC{l_2}$$ | $$N{H_3}$$ | $$B{r_2}/KOH$$ |
| (c) | $$HN{O_2}$$ | $$P + {I_2}$$ | $$LiAl{H_4}$$ |
| (d) | $$HCN$$ | $$N{H_3}$$ | $$B{r_2}/KOH$$ |
A
(a)
B
(b)
C
(c)
D
(d)
Answer :
(b)
130.
The correct order of basicities of the following compounds is
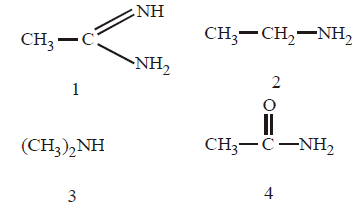
A
2 > 1 > 3 > 4
B
1 > 3 > 2 > 4
C
3 > 1 > 2 > 4
D
1 > 2 > 3 > 4
Answer :
1 > 3 > 2 > 4