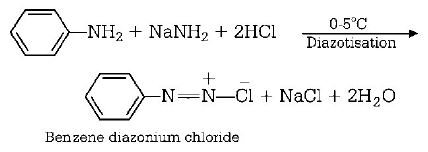
151. Method by which aniline cannot be prepared is
A
hydrolysis phenyl isocyanide with acidic solution
B
degradation of benzamide with bromine in alkaline solution
C
reduction of nitrobenzene with $${H_2}/Pd$$ in ethanol
D
potassium salt of phthalimide treated with chlorobenzene followed by the hydrolysis with aqueous $$NaOH$$ solution
Answer :
potassium salt of phthalimide treated with chlorobenzene followed by the hydrolysis with aqueous $$NaOH$$ solution
152.
Which of the following species are involved in the carbylamine test?
$$\eqalign{
& \left( {\text{i}} \right)RNC \cr
& \left( {{\text{ii}}} \right)CHC{l_3} \cr
& \left( {{\text{iii}}} \right)COC{l_2} \cr
& \left( {{\text{iv}}} \right)NaN{O_2} + HCl \cr} $$
A
(i) and (iv) only
B
(i) and (ii) only
C
(ii) and (iv) only
D
(ii) and (iii) only
Answer :
(i) and (ii) only
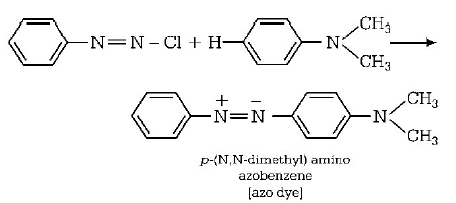
153. Aniline when diazotised in cold and then treated with dimethyl aniline, gives a coloured product. Its structure would be
A
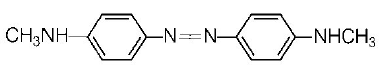
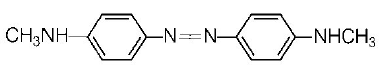
B
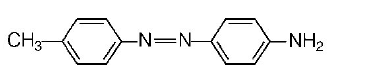

C
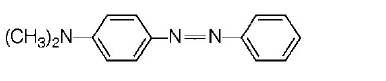
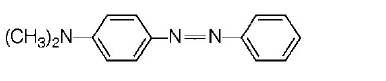
D
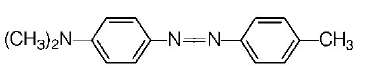
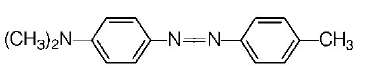
Answer :
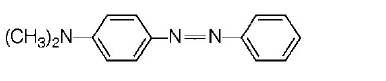
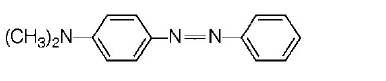
154. Which of the following compounds cannot be identified by carbylamine test?
A
$$C{H_3}C{H_2}N{H_2}$$
B
$${\left( {C{H_3}} \right)_2}CHN{H_2}$$
C
$${C_6}{H_5}N{H_2}$$
D
$${C_6}{H_5}NH{C_6}{H_5}$$
Answer :
$${C_6}{H_5}NH{C_6}{H_5}$$
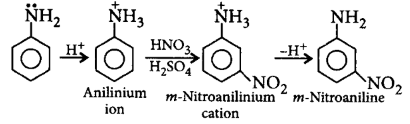
155. Amino group is $$o, p$$ -directing for electrophilic substitution reaction. But, on nitration the major product is $$m$$-nitroaniline because
A
aniline gets protonated with strong acids to give anilinium ion which is $$m$$-directing
B
nitration requires nitric acid which oxidises $$ - N{H_2}$$ to $$ - N{O_2}$$ group
C
electrophile $$NO_2^ + $$ is a $$m$$-directing group
D
benzene ring exerts $$ + I$$ effect and deactivates the ring
Answer :
aniline gets protonated with strong acids to give anilinium ion which is $$m$$-directing
156.
The correct decreasing order of basic strength of the following species is ________.
$${H_2}O,N{H_3},O{H^ - },NH_2^ - $$
A
$$NH_2^ - > O{H^ - } > N{H_3} > {H_2}O$$
B
$$O{H^ - } > NH_2^ - > {H_2}O > N{H_3}$$
C
$$N{H_3} > {H_2}O > NH_2^ - > O{H^ - }$$
D
$${H_2}O > N{H_3} > O{H^ - } > NH_2^ - $$
Answer :
$$NH_2^ - > O{H^ - } > N{H_3} > {H_2}O$$
157. Nitrogen atom of amino group is ________ hybridised.
A
$$sp$$
B
$$s{p^2}$$
C
$$s{p^3}$$
D
$$s{p^3}d$$
Answer :
$$s{p^3}$$
158. In diazotisation reaction of aniline with $$NaN{O_2}$$ and $$HCl,$$ an excess of $$HCl$$ is used to
A
suppress the formation of acetanilide
B
suppress the concentration of free aniline available for coupling
C
maintain the $$pH$$ of the reaction
D
prevent the formation of anilinium ion
Answer :
suppress the concentration of free aniline available for coupling
159. Ethyl isocyanide on hydrolysis in acidic medium generates
A
propanoic acid and ammonium salt
B
ethanoic acid and ammonium salt
C
methylamine salt and ethanoic acid
D
ethylamine salt and methanoic acid
Answer :
ethylamine salt and methanoic acid
160.
\[C{{H}_{3}}-C{{H}_{2}}C\equiv N\xrightarrow{X}C{{H}_{3}}C{{H}_{2}}CHO.\]
The compound $$X$$ is
A
\[SnC{{l}_{2}}/HCl/{{H}_{2}}O,\text{boil}\]
B
\[{{H}_{2}}/Pb-BaS{{O}_{4}}\]
C
\[LiAI{{H}_{4}}/\text{ether}\]
D
\[NaB{{H}_{4}}/\text{ether}/{{H}_{3}}{{O}^{+}}\]
Answer :
\[SnC{{l}_{2}}/HCl/{{H}_{2}}O,\text{boil}\]