201. Acetaldoxime reacts with \[{{P}_{2}}{{O}_{5}}\] to give
A
Methyl cyanide
B
Methyl cyanate
C
Ethyl cyanide
D
None of these
Answer :
Methyl cyanide
202.
Arrange the following amines in the decreasing order of basicity :
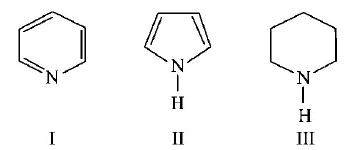
A
I > II > III
B
III > I > II
C
III > II > I
D
I > III > II
Answer :
III > I > II
203. The reaction \[\overset{+\,\,\,}{\mathop{Ar{{N}_{2}}C{{l}^{-}}}}\,\xrightarrow{Cu/HCl}ArCl+{{N}_{2}}\] \[+\,CuCl\] is named as _________.
A
Sandmeyer reaction
B
Gatterman reaction
C
Claisen reaction
D
Carbylamine reaction
Answer :
Gatterman reaction
204. Which of the following has highest $$p{K_b}$$ value?
A
$${\left( {C{H_3}} \right)_3}CN{H_2}$$
B
$$N{H_3}$$
C
$${\left( {C{H_3}} \right)_2}NH$$
D
$$C{H_3}N{H_2}$$
Answer :
$$N{H_3}$$
205. For nitration of aniline, which of the following steps is followed?
A
Direct nitration using nitrating mixture $$\left( {{\text{conc}}{\text{.}}\,HN{O_3} + {\text{conc}}{\text{.}}\,{H_2}S{O_4}} \right)$$ followed by oxidation.
B
Using fuming $$HN{O_3}$$ carrying out reaction at $$273\,K$$ followed by hydrolysis.
C
Using $$NaN{O_2}$$ and $$HCl$$ followed by reaction with conc. $$HN{O_3}$$ followed by hydrolysis.
D
Acetylation followedby nitration and hydrolysis.
Answer :
Acetylation followedby nitration and hydrolysis.
206.
The correct increasing order of basic strength for the following compounds is
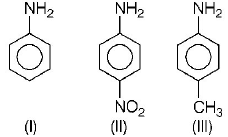
A
II < III < I
B
III < I < II
C
III < II < I
D
II < I < III
Answer :
II < I < III
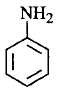
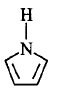
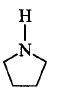
207. Among the following amines, the strongest $${\rm{Br\ddot onsted}}$$ base is ________.
A

B
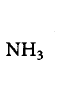
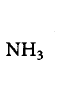
C

D

Answer :


208. The most basic amine among the following is
A
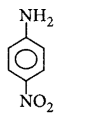

B
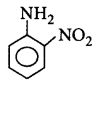

C
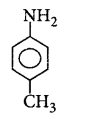

D
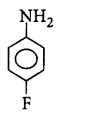

Answer :


209.
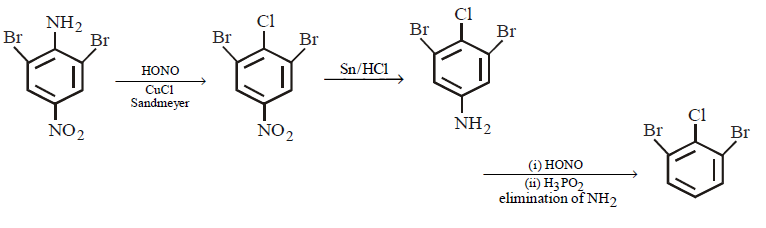
The product – $$(C)$$ obtained in the following sequence of reactions is
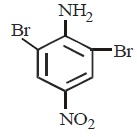 \[\xrightarrow[\left( 2 \right)\,CuCl]{\left( 1 \right)\,HONO}A\xrightarrow{Sn/HCl}B\xrightarrow[\left( 2 \right)\,{{H}_{3}}P{{O}_{2}}]{\left( 1 \right)\,HONO}C\]
\[\xrightarrow[\left( 2 \right)\,CuCl]{\left( 1 \right)\,HONO}A\xrightarrow{Sn/HCl}B\xrightarrow[\left( 2 \right)\,{{H}_{3}}P{{O}_{2}}]{\left( 1 \right)\,HONO}C\]
A
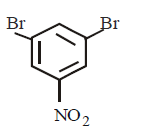

B
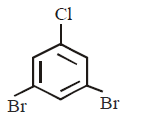

C
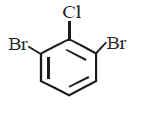

D
Answer :


210.
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}\xrightarrow{Sn/HCl}P\xrightarrow[HCl]{NaN{{O}_{2}}}\] \[Q\xrightarrow[\Delta ]{HB{{F}_{4}}}R\]
The end product $$R$$ in the given sequence of reactions is
A
benzoic acid
B
fluorobenzene
C
phenol
D
chlorobenzene
Answer :
fluorobenzene
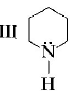 is most basic as the lone pair of nitrogen is easily available for the donation.
is most basic as the lone pair of nitrogen is easily available for the donation.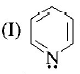 lone pair is not involved in resonance but nitrogen atom is $$S{p^2}$$ hybridsed whereas in compound II the lone pair of nitrogen is involved in aromaticity which makes it least basic.
lone pair is not involved in resonance but nitrogen atom is $$S{p^2}$$ hybridsed whereas in compound II the lone pair of nitrogen is involved in aromaticity which makes it least basic. 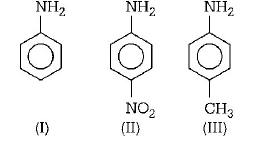
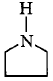 is the strongest $${\rm{Br\ddot onsted}}$$ base as there is no delocalisation of lone pair of electrons of $$N$$ atom which is possible in aniline and in pyrrole.
is the strongest $${\rm{Br\ddot onsted}}$$ base as there is no delocalisation of lone pair of electrons of $$N$$ atom which is possible in aniline and in pyrrole.