211.
Arrange the following three compounds in order of decreasing acidity
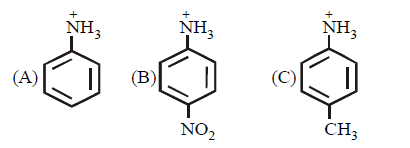
A
B > A > C
B
B > C > A
C
C > B > A
D
C > A > B
Answer :
B > A > C
212. An organic compound $$(X)$$ was treated with sodium nitrite and $$HCl$$ in ice cold conditions. Bubbles of nitrogen gas were seen coming out. The compound $$(X)$$ may be
A
a secondary aliphatic amine
B
a primary aromatic amine
C
a primary aliphatic amine
D
a tertiary amine
Answer :
a primary aliphatic amine
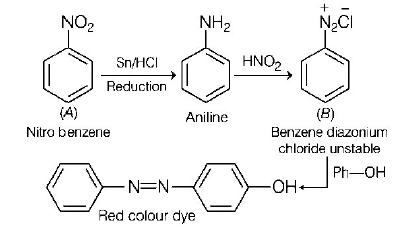
213. A given nitrogen-containing aromatic compound $$A$$ reacts with $$Sn/HCl,$$ followed by $$HN{O_2}$$ to give an unstable compound $$B.$$ $$B,$$ on treatment with phenol, forms a beautify coloured compound $$C$$ with the molecular formula $${C_{12}}{H_{10}}{N_2}O.$$ The structure of compound $$A$$ is
A
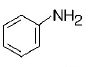

B
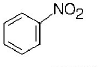

C
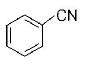

D
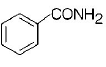

Answer :


214.
In the following reaction,
$$C{H_3}N{H_2} + CHC{l_3} + KOH \to $$ $${\text{Nitrogen containing compound}}$$ $$ + KCl + {H_2}O.$$
The nitrogen containing compound is
A
$$C{H_3} - NH - C{H_3}$$
B
$$C{H_3} - C \equiv N$$
C
$$C{H_3} - \mathop N\limits^ + \equiv \mathop C\limits^ - $$
D
$$C{H_3} - \mathop N\limits^ - \equiv \mathop C\limits^ + $$
Answer :
$$C{H_3} - \mathop N\limits^ + \equiv \mathop C\limits^ - $$
215.
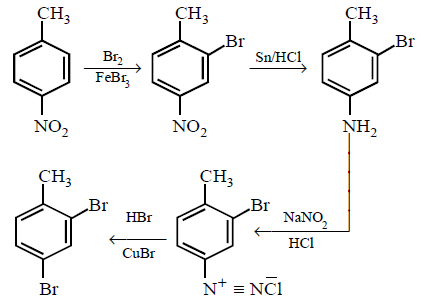
In a set of reactions $$p$$ - nitrotoluene yielded a product $$E.$$
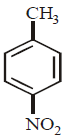 \[\xrightarrow[FeB{{r}_{3}}]{B{{r}_{2}}}B\xrightarrow{Sn/HCl}C\xrightarrow[HCl]{NaN{{O}_{2}}}D\xrightarrow[HBr]{CuBr}E\]
\[\xrightarrow[FeB{{r}_{3}}]{B{{r}_{2}}}B\xrightarrow{Sn/HCl}C\xrightarrow[HCl]{NaN{{O}_{2}}}D\xrightarrow[HBr]{CuBr}E\]
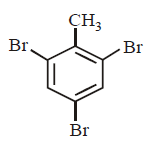
The product $$E$$ would be :
A

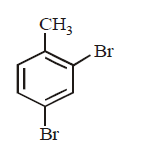
B

C
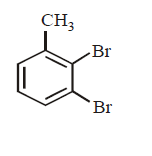

D
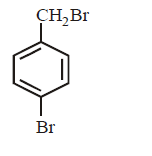

Answer :


216. Benzenediazonium chloride cannot be stored and is used immediately after its preparation because
A
it slowly evaporates on storage
B
it is very unstable and dissociates to give nitrogen
C
it gets oxidised in air hence cannot be stored
D
it reacts with all the containers in which it is stored
Answer :
it is very unstable and dissociates to give nitrogen
217.
In the acidic reduction of nitrobenzene, which of the following is the intermediate ?
A
$${C_6}{H_5} - N = O$$
B
$${C_6}{H_5}NH - NH{C_6}{H_5}$$
C
$${C_6}{H_5} - N = N - {C_6}{H_5}$$
D
\[{{C}_{6}}{{H}_{5}}-N=\overset{\begin{smallmatrix}
O \\
\uparrow
\end{smallmatrix}}{\mathop{N}}\,-{{C}_{6}}{{H}_{5}}\]
Answer :
$${C_6}{H_5} - N = O$$
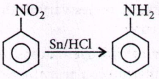
218. Reduction of aromatic nitro compounds using $$Sn$$ and $$HCl$$ gives
A
aromatic primary amines
B
aromatic secondary amines
C
aromatic tertiary amines
D
aromatic amides
Answer :
aromatic primary amines
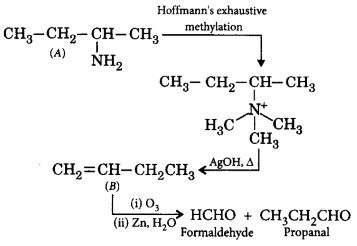
219. When an optically active amine $$(A)$$ having molecular formula $${C_4}{H_{11}}N$$ is subjected to Hoffmann's exhaustive methylation followed by hydrolysis, an alkene $$(B)$$ is produced which upon ozonolysis and subsequent hydrolysis yields formaldehyde and propanal. The amine $$'A'$$ is
A
\[\underset{\begin{smallmatrix}
|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\, \\
N{{H}_{2}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,
\end{smallmatrix}}{\mathop{C{{H}_{3}}CHC{{H}_{2}}C{{H}_{3}}}}\,\]
B
\[\underset{\begin{smallmatrix}
\,\,\,\,| \\
\,\,\,\,\,\,\,C{{H}_{3}}
\end{smallmatrix}}{\mathop{C{{H}_{3}}NH-CH-C{{H}_{3}}}}\,\]
C
\[\underset{\begin{smallmatrix}
|\,\,\,\,\,\,\,\,\,\,\,\,\,\, \\
C{{H}_{3}}\,\,\,\,\,\,\,\,\,\,
\end{smallmatrix}}{\mathop{C{{H}_{3}}NC{{H}_{2}}C{{H}_{3}}}}\,\]
D
$$C{H_3}C{H_2}C{H_2}C{H_2}N{H_2}$$
Answer :
\[\underset{\begin{smallmatrix}
|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\, \\
N{{H}_{2}}\,\,\,\,\,\,\,\,\,\,\,\,\,\,
\end{smallmatrix}}{\mathop{C{{H}_{3}}CHC{{H}_{2}}C{{H}_{3}}}}\,\]
220.
In the compound given below the correct order of the acidity of the positions $$X,Y$$ and $$Z$$ is
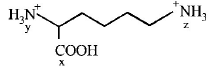
A
$$Z > X > Y$$
B
$$X > Y > Z$$
C
$$X > Z > Y$$
D
$$Y > X > Z$$
Answer :
$$X > Y > Z$$