131. $${I_2}\left( s \right)\left| {{I^ - }\left( {0.1\,M} \right)} \right.$$ half cell is connected to a $${H^ + }\left( {aq} \right)\left| {{H_2}\left( {1\,bar} \right)} \right|Pt$$ half cell and $$e.m.f.$$ is found to be $$0.7714\,V.$$ if $$E_{{I_2}\left| {{I^ - }} \right.}^ \circ = 0.535\,V,$$ find the $$pH$$ of $${H^ + }\left| {{H_2}} \right.$$ half-cell.
A
1
B
3
C
5
D
7
Answer :
3
132.
The equivalent conductivity of $$\frac{N}{{10}}$$ solution of acetic acid at $${25^ \circ }C$$ is $$14.3\,oh{m^{ - 1}}\,c{m^2}\,equi{v^{ - 1}}.$$ What will be the degree of dissociation of acetic acid?
$$\left( {{ \wedge _{\infty \,\,C{H_3}COOH}} = 390.71\,oh{m^{ - 1}}\,c{m^2}\,equi{v^{ - 1}}} \right)$$
A
3.66 %
B
4.9 %
C
2.12 %
D
0.008 %
Answer :
3.66 %
133. Which of the following reaction is possible at anode?
A
$$2C{r^{3 + }} + 7{H_2}O \to C{r_2}{O_7}^{2 - } + 14{H^ + }$$
B
$${F_2} \to 2{F^ - }$$
C
$$\left( {\frac{1}{2}} \right){O_2} + 2{H^ + } \to {H_2}O$$
D
None of these.
Answer :
$$2C{r^{3 + }} + 7{H_2}O \to C{r_2}{O_7}^{2 - } + 14{H^ + }$$
134.
The $$e.m.f.$$ of a Daniell cell at $$298\,K$$ is $${E_1}.$$
$$Zn\left| {\mathop {ZnS{O_4}}\limits_{\left( {0.01\,M} \right)} } \right|\left| {\mathop {CuS{O_4}}\limits_{\left( {1.0\,M} \right)} } \right|Cu$$
When the concentration of $${ZnS{O_4}}$$ is $$1.0\,M$$ and that of $${CuS{O_4}}$$ is $$0.01\,M,$$ the $$e.m.f.$$ changed to $${E_2}.$$ What is the relationship between $${E_1}$$ and $${E_2}?$$
A
$${E_2} = 0 \ne {E_1}$$
B
$${E_1} > {E_2}$$
C
$${E_1} < {E_2}$$
D
$${E_1} = {E_2}$$
Answer :
$${E_1} > {E_2}$$
135. $${E^ \circ }$$ value of $$\frac{{N{i^{2 + }}}}{{Ni}}$$ is $$ - 0.25\,V$$ and $$\frac{{A{g^ + }}}{{Ag}}$$ is $$ + 0.80\,V.$$ If a cell is made by taking the two electrodes what is the feasibility of the reaction?
A
Since $${E^ \circ }$$ value for the cell will be positive, redox reaction is feasible.
B
Since $${E^ \circ }$$ value for the cell will be negative, redox reaction is not feasible.
C
$$Ni$$ cannot reduce $${A{g^ + }}$$ to $$Ag$$ hence reaction is not feasible.
D
$$Ag$$ can reduce $${N{i^{2 + }}}$$ to $$Ni$$ hence reaction is feasible.
Answer :
Since $${E^ \circ }$$ value for the cell will be positive, redox reaction is feasible.
136. Which one of the following pairs of substances on reaction will not evolve $${H_2}$$ gas?
A
Iron and $${H_2}S{O_4}\left( {aq} \right)$$
B
Iron and steam
C
Copper and $$HCl\left( {aq} \right)$$
D
Sodium and ethyl alcohol
Answer :
Copper and $$HCl\left( {aq} \right)$$
137.
$$\eqalign{
& { \wedge _{ClC{H_2}COONa}} = 224\,oh{m^{ - 1}}\,c{m^2}g\,e{q^{ - 1}}, \cr
& { \wedge _{NaCl}} = 38.2\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}, \cr
& { \wedge _{HCl}} = 203\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}, \cr} $$
What is the value of $${ \wedge _{ClC{H_2}COOH}}$$
A
$$288.5\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}$$
B
$$289.5\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}$$
C
$$388.8\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}$$
D
$$59.5\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}$$
Answer :
$$388.8\,oh{m^{ - 1}}c{m^2}g\,e{q^{ - 1}}$$
138.
The Gibbs energy for the decomposition of $$A{{\text{l}}_2}{O_3}$$ at $${500^ \circ }C$$ is as follows :
$$\frac{2}{3}A{{\text{l}}_2}{O_3} \to \frac{4}{3}A{\text{l}} + {O_2},{\Delta _r}G = + 966\,kJ\,mo{l^{ - 1}}$$
The potential difference needed for electrolytic reduction of $$A{{\text{l}}_2}{O_3}$$ at $${500^ \circ }C$$ is at least
A
$$4.5 V$$
B
$$3.0 V$$
C
$$2.5 V$$
D
$$5.0 V$$
Answer :
$$2.5 V$$
139.
Which of the following is the correct cell representation for the given cell reaction?
$$Zn + {H_2}S{O_4} \to ZnS{O_4} + {H_2}$$
A
$$Zn\left| {Z{n^{2 + }}} \right|\left| {{H^ + }} \right|{H_2}$$
B
$$Zn\left| {Z{n^{2 + }}} \right|\left| {{H^ + },{H_2}} \right|Pt$$
C
$$Zn\left| {ZnS{O_4}} \right|\left| {{H_2}S{O_4}} \right|Zn$$
D
$$Zn\left| {{H_2}S{O_4}} \right|\left| {ZnS{O_4}} \right|{H_2}$$
Answer :
$$Zn\left| {Z{n^{2 + }}} \right|\left| {{H^ + },{H_2}} \right|Pt$$
140. $$HN{O_3}\left( {aq} \right)$$ is titrated with $$NaOH\left( {aq} \right)$$ conductometrically, graphical representation of the titration is :
A
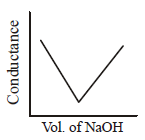

B
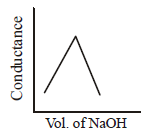

C


D
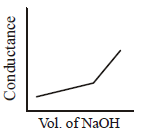

Answer :

