61. The efficiency of a fuel cell is given by
A
$$\frac{{\Delta G}}{{\Delta S}}$$
B
$$\frac{{\Delta G}}{{\Delta H}}$$
C
$$\frac{{\Delta S}}{{\Delta G}}$$
D
$$\frac{{\Delta H}}{{\Delta G}}$$
Answer :
$$\frac{{\Delta G}}{{\Delta H}}$$
62.
Which of the given statements for mercury cell are incorrect?
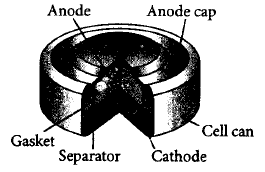
(i) Mercury cell is suitable for low current devices like hearing aids, watches, etc.
(ii) It consists of zinc-mercury amalgam as anode and a paste of $$HgO$$ and carbon as the cathode.
(iii) The electrolyte is a paste of $$Zn{\left( {OH} \right)_2}$$ and $$K{O_2}.$$
(iv) The electrode reactions for the cell are
At anode : $$Zn\left( {Hg} \right) + {H_2}O \to $$ $$Zn{O_{\left( s \right)}} + 2O{H^ - } + 2{e^ - }$$
At cathode : $$HgO + {H_2}O + 2{e^ - } \to $$ $$H{g_{\left( l \right)}} + 2O{H^ - }$$
A
(ii) and (iii) only
B
(i) and (ii) only
C
(i), (iii) and (iv) only
D
(iii) and (iv) only
Answer :
(iii) and (iv) only
63. An unknown metal M displaces nickel from nickel (II) sulphate solution but does not displace manganese from manganese sulphate solution. Which order represents the correct order of reducing power?
A
$$Mn > Ni > M$$
B
$$Ni > Mn > M$$
C
$$Mn > M > Ni$$
D
$$M > Ni > Mn$$
Answer :
$$Mn > M > Ni$$
64. In a cell that utilises the reaction $$Z{n_{\left( s \right)}} + 2{H^ + }_{\left( {aq} \right)} \to Z{n^{2 + }}_{\left( {aq} \right)} + {H_{2\left( g \right)}}$$ addition of $${H_2}S{O_4}$$ to cathode compartment, will
A
increase the $$E$$ and shift equilibrium to the right
B
lower the $$E$$ and shift equilibrium to the right
C
lower the $$E$$ and shift equlibrium to the left
D
increase the $$E$$ and shift equilibrium to the left
Answer :
increase the $$E$$ and shift equilibrium to the right
65.
Label the given diagram showing lead storage battery :
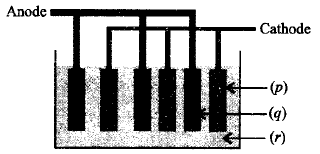
| $$p$$ | $$q$$ | $$r$$ | |
| (a) | $$Pb$$ | $$Pb{O_2}$$ | $$5\,M\,{H_2}S{O_4}$$ |
| (b) | $$Pb{O_2}$$ | $$Pb$$ | $$conc.\,{H_2}S{O_4}$$ |
| (c) | $$P{b_3}{O_4}$$ | $$Pb{O_2}$$ | $$50\% \,{H_2}S{O_4}$$ |
| (d) | $$Pb{O_2}$$ | $$Pb$$ | $$dil.\,38\% \,{H_2}S{O_4}$$ |
A
(a)
B
(b)
C
(c)
D
(d)
Answer :
(d)
66. When a lead storage battery is discharged,
A
lead sulphate is consumed
B
oxygen gas is evolved
C
lead sulphate is formed
D
lead sulphide is formed
Answer :
lead sulphate is formed
67.
For the reduction of silver ions with copper metal, the standard cell potential was found to be $$+ 0.46$$ $$V$$ at $${25^ \circ }C.$$ The value of standard Gibbs energy, $$\Delta {G^ \circ }$$ will be
$$\left( {F = 96500\,C\,mo{l^{ - 1}}} \right)$$
A
$$ - 89.0\,kJ$$
B
$$ - 89.0\,J$$
C
$$ - 44.5\,kJ$$
D
$$ - 98.0\,kJ$$
Answer :
$$ - 89.0\,kJ$$
68. The charge required for reducing $$1\,mole$$ of $$MnO_4^ - $$ to $$M{n^{2 + }}$$ is
A
$$1.93 \times {10^5}\,C$$
B
$$2.895 \times {10^5}\,C$$
C
$$4.28 \times {10^5}\,C$$
D
$$4.825 \times {10^5}\,C$$
Answer :
$$4.825 \times {10^5}\,C$$
69. The standard electrode potential $$\left( {{E^ \circ }} \right)$$ for $$\frac{{OC{l^ - }}}{{C{l^ - }}}$$ and $$\frac{1}{2}\frac{{C{l^ - }}}{{C{l_2}}}$$ respectively are $$0.94\,V$$ and $$ - 1.36\,V.$$ The $${E^ \circ }$$ value for $$\frac{1}{2}\frac{{OC{l^ - }}}{{C{l_2}}}$$ will be
A
$$- 0.42\,V$$
B
$$- 2.20\,V$$
C
$$0.52\,V$$
D
$$1.04\,V$$
Answer :
$$- 0.42\,V$$
70. $$Zn$$ gives $${H_2}$$ gas with $${H_2}S{O_4}$$ and $$HCl$$ but not with $$HN{O_3}$$ because
A
$$Zn$$ acts as oxidizing when reacts with $$HN{O_3}.$$
B
$$HN{O_3}.$$ is weaker acid than $${H_2}S{O_4}$$ and $$HCl.$$
C
In electrochemical series $$Zn$$ is above hydrogen.
D
$$NO_3^ - $$ is reduced in preference to hydronium.
Answer :
$$NO_3^ - $$ is reduced in preference to hydronium.