91. Non-stoichiometric compound have the properties of :
A
Electrical conductance
B
Isolation
C
Insulation
D
None of these
Answer :
None of these
92. In $$ABC\,ABC$$ packing, if the number of atoms in the unit cell is $$n$$ then the number of tetrahedral voids in the unit cell is equal to
A
$$n$$
B
$$\frac{n}{2}$$
C
$$\frac{n}{4}$$
D
$$2n$$
Answer :
$$2n$$
93. The radii of $$N{a^ + }$$ and $$C{l^ - }\,ions$$ are $$95\,pm$$ and $$181\,pm$$ respectively. The edge length of $$NaCl$$ unit cell is
A
276$$\,pm$$
B
138$$\,pm$$
C
552$$\,pm$$
D
415$$\,pm$$
Answer :
552$$\,pm$$
94.
What type of crystal defect is indicated in the diagram below?
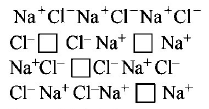
A
Interstitial defect
B
Schottky defect
C
Frenkel defect
D
Frenkel and Schottky defects .
Answer :
Schottky defect
95. If we mix a pentavalent impurity in a crystal lattice of germanium, what type of semiconductor formation will occur?
A
$$p$$-type
B
$$n$$-type
C
Both (A) and (B)
D
Neither (A) nor (B)
Answer :
$$n$$-type
96. $$NaCl$$ type crystal ( with coordination no. 6 : 6 ) can be converted into $$CsCl$$ type crystal ( with coordination no. 8 : 8 ) by applying
A
high temperature
B
high pressure
C
high temperature and high pressure
D
low temperature and low pressure
Answer :
high pressure
97. Which of the following crystals does not exhibit Frenkel defect ?
A
$$AgBr$$
B
$$AgCl$$
C
$$KBr$$
D
$$ZnS$$
Answer :
$$KBr$$
98. Percentages of free space in cubic close packed structure and in body centered packed structure are respectively
A
30% and 26%
B
26% and 32%
C
32% and 48%
D
48% and 26%
Answer :
26% and 32%
99. To get $$n$$ - type of semiconductor, germanium should be doped with
A
gallium
B
arsenic
C
aluminium
D
boron
Answer :
arsenic
100.
Which of the following solids is not an electrical conductor ?
$$\eqalign{
& \left( {\text{i}} \right)M{g_{\left( s \right)}} \cr
& \left( {{\text{ii}}} \right)Ti{O_{\left( s \right)}} \cr
& \left( {{\text{iii}}} \right){I_{2\left( s \right)}} \cr
& \left( {{\text{iv}}} \right){H_2}{O_{\left( s \right)}} \cr} $$
A
(i) only
B
(ii) only
C
(iii) and (iv)
D
(ii), (iii) and (iV)
Answer :
(iii) and (iv)