121.
Which of the following primitive cells show the given parameters ?
$$a \ne b \ne c,\,\,\alpha = \beta = \gamma = {90^ \circ }$$
A
Cubic
B
Tetragonal
C
Orthorhombic
D
Hexagonal
Answer :
Orthorhombic
122. The correct order of the packing efficiency in different types of unit cells is _________.
A
$$fcc < bcc < {\text{simple cubic}}$$
B
$$fcc > bcc > {\text{simple cubic}}$$
C
$$fcc < bcc > {\text{simple cubic}}$$
D
$$bcc < fcc > {\text{simple cubic}}$$
Answer :
$$fcc > bcc > {\text{simple cubic}}$$
123. How many unit cells are present in $$5.0\,gm$$ of crystal $$AB$$ ( formula mass of $$AB = 40$$ ) having rock salt type structure ? ( $${N_A} = $$ Avogadro's no. )
A
$${N_A}$$
B
$$\frac{{{N_A}}}{{10}}$$
C
$$4\,{N_A}$$
D
$${\text{none of these}}$$
Answer :
$${\text{none of these}}$$
124. Which of the following exists as covalent crystals in the solid state ?
A
lodine
B
Silicon
C
Sulphur
D
Phosphorus
Answer :
Silicon
125. A metal crystallises into two cubic phases, face centred cubic $$(fcc)$$ and body centred cubic $$(bcc),$$ whose unit cell lengths are $$3.5\mathop {\text{A}}\limits^{\text{o}} $$ and $$3.0\mathop {\text{A}}\limits^{\text{o}} ,$$ respectively. The ratio of densities of $$fcc$$ and $$bcc$$ is
A
1.259 : 1
B
1 : 1.259
C
3 : 2
D
1.142 : 1
Answer :
1.259 : 1
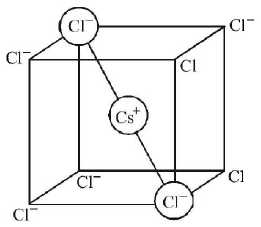
126. $$CsCl$$ crystallises in body centred cubic lattice. If $$‘a’$$ is its edge length then which of the following expressions is correct?
A
$${r_{Cs}} + {r_{C{l^ - }}} = 3a$$
B
$${r_{C{s^ + }}} + {r_{C{l^ - }}} = \frac{{3a}}{2}$$
C
$${r_{C{s^ + }}} + {r_{C{l^ - }}} = \frac{{\sqrt 3 }}{2}a$$
D
$${r_{C{s^ + }}} + {r_{C{l^ - }}} = \sqrt {3a} $$
Answer :
$${r_{C{s^ + }}} + {r_{C{l^ - }}} = \frac{{\sqrt 3 }}{2}a$$
127. In the solid state, $$MgO$$ has the same structure as that of sodium chloride. The number of oxygens surrounding each magnesium in $$MgO$$ is
A
6
B
1
C
2
D
4
Answer :
6
128. Examples of few solids are given below. Find out the example which is not correctly matched.
A
Ionic solids - $$NaCl,ZnS$$
B
Covalent solids - $${H_2},{I_2}$$
C
Molecular solids - $${H_2}{O_{\left( s \right)}}$$
D
Metallic solids - $$Cu,Sn$$
Answer :
Covalent solids - $${H_2},{I_2}$$
129. Total volume of atoms present in a face-centred cubic unit cell of a metal is ( $$r$$ is atomic radius )
A
$$\frac{{12}}{3}\pi {r^3}$$
B
$$\frac{{16}}{3}\pi {r^3}$$
C
$$\frac{{20}}{3}\pi {r^3}$$
D
$$\frac{{24}}{3}\pi {r^3}$$
Answer :
$$\frac{{16}}{3}\pi {r^3}$$
130.
Which of the following shows correct range of conductivity ?
(i) Conductors : $${10^4}\,\,{\text{to}}\,\,{10^7}oh{m^{ - 1}}{m^{ - 1}}$$
(ii) Insulators : $${10^{ - 6}}\,\,{\text{to}}\,\,{10^4}oh{m^{ - 1}}{m^{ - 1}}$$
(iii) Semiconductors : $${10^{ - 10}}\,\,{\text{to}}\,\,{10^{ - 6}}oh{{\dot m}^{ - 1}}{m^{ - 1}}$$
A
(i) and (ii) only
B
(i) only
C
(ii) and (iii) only
D
(i), (ii) and (iii)
Answer :
(i) only