131. An organic compound with the formula $${C_6}{H_{12}}{O_6}$$ forms a yellow crystalline solid with phenylhydrazine and gives a mixture of sorbitol and mannitol when reduced with sodium. Which among the following could be the compound ?
A
fructose
B
glucose
C
mannose
D
sucrose
Answer :
fructose
132. Synthesis of each molecule of glucose in photosynthesis involves :
A
18 molecules of $$ATP$$
B
10 molecules of $$ATP$$
C
8 molecules of $$ATP$$
D
6 molecules of $$ATP$$
Answer :
18 molecules of $$ATP$$
133. $$DNA$$ and $$RNA$$ contain four bases each. Which of the following bases is not present in $$RNA?$$
A
Adenine
B
Uracil
C
Thymine
D
Cytosine
Answer :
Thymine
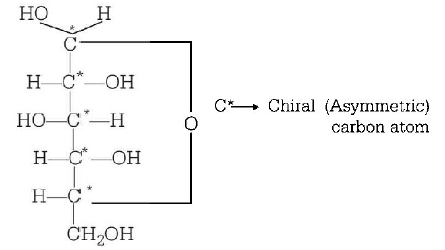
134. Number of chiral carbon atoms in $$\beta - D - \left( + \right) - $$ glucose is
A
five
B
six
C
three
D
four
Answer :
five
135. Carbohydrates are stored in human body as the polysaccharide
A
starch
B
glycogen
C
cellulose
D
amylose
Answer :
glycogen
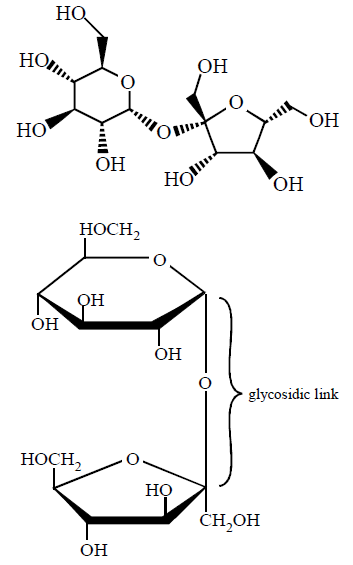
136. Glycosidic linkage is actually an :
A
Carbonyl bond
B
Ether bond
C
Ester bond
D
Amide bond
Answer :
Ether bond
137. Vegetable oils like wheat germ oil, sunflower oil, etc. are the good source of
A
vitamin $$K$$
B
vitamin $$E$$
C
vitamin $$D$$
D
vitamin $$A$$
Answer :
vitamin $$E$$
138. The segment of $$DNA$$ which acts as the instrumental manual for the synthesis of the protein is
A
nucleotide
B
ribose
C
gene
D
nucleoside
Answer :
gene
139. Benedict's solution is used to identify
A
lipids
B
proteins
C
reducing sugars
D
polysaccharide
Answer :
reducing sugars
140. Sucrose in water is dextrorotatory, $${\left[ \alpha \right]_D} = + {66.4^ \circ }$$ when boiled with dil. $$HCl,$$ the solution becomes leavorotatory, $${\left[ \alpha \right]_D} = - {39.9^ \circ }.$$ In this process the sucrose breaks into
A
$$L$$ - glucose + $$D$$ - fructose
B
$$L$$ - glucose + $$L$$ - fructose
C
$$D$$ - glucose + $$D$$ - fructose
D
$$D$$ - glucose + $$L$$ - fructose
Answer :
$$D$$ - glucose + $$D$$ - fructose