61. Which one of the following is an amine hormone ?
A
Thyroxine
B
Oxypurin
C
Insulin
D
Progesterone
Answer :
Thyroxine
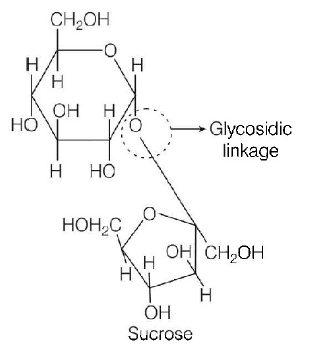
62. Which one given below is a non-reducing sugar?
A
Lactose
B
Glucose
C
Sucrose
D
Maltose
Answer :
Sucrose
63. Which one of the amino acids can be synthesised in the body?
A
Alanine
B
Lysine
C
Valine
D
Histidine
Answer :
Alanine
64. Amino acids generally exist in the form of Zwitter ions. This means they contain
A
basic $$ - N{H_2}$$ group and acidic $$-COOH$$ group
B
the basic $$\mathop { - N{H_3}}\limits^{ + \,} $$ group and acidic $$ - CO{O^ - }$$ group
C
basic $$ - N{H_2}$$ and acidic $$ - {H^ + }$$ group
D
basic $$ - CO{O^ - }$$ group and acidic $$\mathop { - N{H_3}}\limits^{ + \,} $$ group
Answer :
basic $$ - CO{O^ - }$$ group and acidic $$\mathop { - N{H_3}}\limits^{ + \,} $$ group
65. Proteins are condensation polymers of
A
$$\alpha $$ - amino acids
B
$$\beta $$ - amino acids
C
$$\alpha $$ - hydroxy acids
D
$$\beta $$ - hydroxy acids
Answer :
$$\alpha $$ - amino acids
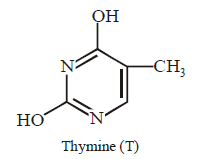
66. Which of the following structures represents thymine ?
A
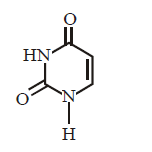

B
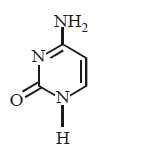

C
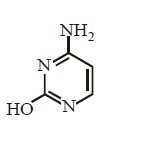

D
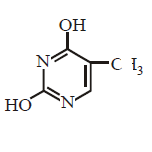

Answer :


67. What will happen when $$D - \left( + \right) - $$ glucose is treated with methanolic $$-HCl$$ followed by Tollens’ reagent ?
A
A black $$ppt.$$ will be formed
B
A red $$ppt.$$ will be formed
C
A green colour will appear
D
No characteristic colour or $$ppt.$$ will be formed.
Answer :
No characteristic colour or $$ppt.$$ will be formed.
68. On oxidation with a mild oxidising agent like $$B{r_2}/{H_2}O,$$ the glucose is oxidised to
A
saccharic acid
B
glucaric acid
C
gluconic acid
D
valerie acid
Answer :
gluconic acid
69. The correct statement in respect of protein haemoglobin is that it
A
functions as a catalyst for biological reactions
B
maintains blood sugar level
C
act as an oxygen carrier in the blood
D
forms antibodies and offers resistance to diseases
Answer :
act as an oxygen carrier in the blood
70. Vitamin $$C$$ must be supplied regularly in diet because
A
it is water soluble hence excreted in urine and can't be stored in the body
B
it is fat soluble hence stored in the body and cannot be used on regular basis
C
it is required in a large amount by the body hence supplied regularly
D
it is water soluble hence used by the body on daily basis and is to be supplied regularly
Answer :
it is water soluble hence excreted in urine and can't be stored in the body
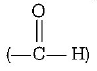 and fructose $$\left( { > C = O} \right)$$ are involved in glycosidic linkage.
and fructose $$\left( { > C = O} \right)$$ are involved in glycosidic linkage.