61. Which of the following will not show geometrical isomerism?
A
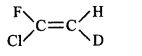
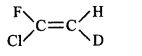
B
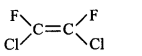
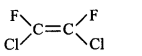
C
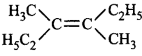
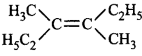
D
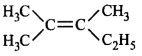
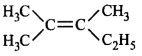
Answer :
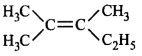
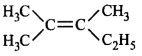
62. Which of the following compounds with molecular formula, $${C_5}{H_{10}}$$ yields acetone on ozonolysis?
A
$$2-methyl-2-butene$$
B
$$3-methyl-1-butene$$
C
$$Cyclopentane$$
D
$$2-methyl-1-butene$$
Answer :
$$2-methyl-2-butene$$
63. The principal organic product formed in the reaction, \[C{{H}_{2}}=CH{{\left( C{{H}_{2}} \right)}_{8}}COOH+HBr\xrightarrow{\text{peroxide}}\] is
A
$$C{H_3}CHBr{\left( {C{H_2}} \right)_8}COOH$$
B
$$C{H_2} = CH{\left( {C{H_2}} \right)_8}COBr$$
C
$$C{H_2}BrC{H_2}{\left( {C{H_2}} \right)_8}COOH$$
D
$$C{H_2} = CH{\left( {C{H_2}} \right)_7}CHBrCOOH$$
Answer :
$$C{H_2}BrC{H_2}{\left( {C{H_2}} \right)_8}COOH$$
64. Which of the following will give 2, 2-dibromopropane on reaction with $$HBr?$$
A
$$C{H_3} - CH = C{H_2}$$
B
$$C{H_3}C \equiv CH$$
C
$$C{H_3}CH = CHBr$$
D
$$CH \equiv CH$$
Answer :
$$C{H_3}C \equiv CH$$
65.
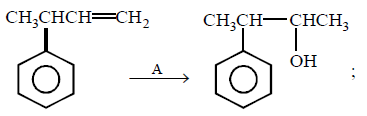
Reagent $$A$$ may be :
A
$${H_2}O/{H^ + }$$
B
$$B{H_3},THF/{H_2}{O_2} - O{H^ - }$$
C
$$Hg{\left( {OCOC{H_3}} \right)_2},{H_2}O/NaB{H_4}.NaOH$$
D
$${\text{All are possible}}$$
Answer :
$$Hg{\left( {OCOC{H_3}} \right)_2},{H_2}O/NaB{H_4}.NaOH$$
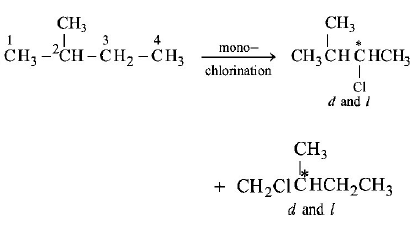
66. On monochlorination of 2 - methylbutane, the total number of chiral compounds formed is
A
2
B
4
C
6
D
8
Answer :
4
67. In the hydroboration - oxidation reaction of propene with diborane, $${H_2}{O_2}$$ and $$NaOH,$$ the organic compound formed is :
A
$$C{H_3}C{H_2}OH$$
B
$$C{H_3}CHOHC{H_3}$$
C
$$C{H_3}C{H_2}C{H_2}OH$$
D
$${\left( {C{H_3}} \right)_3}COH$$
Answer :
$$C{H_3}C{H_2}C{H_2}OH$$
68. Geometrical isomerism is caused
A
by restricted rotation around $$C = C$$ bond
B
by the presence of one asymmetric carbon atom
C
due to the different groups attached to the same functional group
D
by swing of hydrogen atom between two divalent atoms
Answer :
by restricted rotation around $$C = C$$ bond
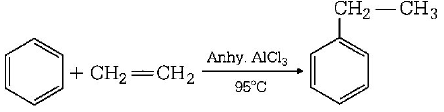
69. Using anhy. $$AlC{l_3}$$ as catalyst, which one of the following reactions produce ethylbenzene $$(PhEt)?$$
A
$${H_3}C - C{H_2}OH + {C_6}{H_6}$$
B
$$C{H_3} - CH = C{H_2} + {C_6}{H_6}$$
C
$${H_2}C = C{H_2} + {C_6}{H_6}$$
D
$${H_3}C - C{H_3} + {C_6}{H_6}$$
Answer :
$${H_2}C = C{H_2} + {C_6}{H_6}$$
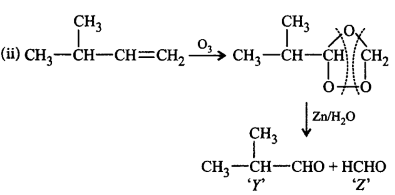
70.
The products for the following reactions are
\[\left( \text{i} \right)C{{H}_{3}}\underset{\begin{smallmatrix}
| \\
H\,
\end{smallmatrix}}{\overset{\begin{smallmatrix}
\,\,\,Br \\
|
\end{smallmatrix}}{\mathop{-C-}}}\,C{{H}_{2}}-C{{H}_{3}}\,\,+\] \[alc.\,KOH\to X\]
\[\left( \text{ii} \right)C{{H}_{3}}\underset{\begin{smallmatrix}
|\,\,\,\,\, \\
C{{H}_{3}}
\end{smallmatrix}}{\mathop{-CH-}}\,CH=C{{H}_{2}}\] \[\xrightarrow{{{O}_{3}}}Y+Z\]
A
$$X = {\left( {C{H_3}} \right)_2}C = C{H_2},$$ $$Y = C{H_3}C{H_2}CHO,$$ $$Z = C{H_3}C{H_2}CHO$$
B
$$X = C{H_2} = C{H_2},$$ $$Y = C{H_3}CHO,$$ $$Z = C{H_3}COOH$$
C
$$X = C{H_3} - CH = CH - C{H_3},$$ \[Y=C{{H}_{3}}\overset{\begin{smallmatrix}
C{{H}_{3}} \\
|\,\,\,\,\,
\end{smallmatrix}}{\mathop{-CH-}}\,CHO,\] $$Z = HCHO$$
D
$$X = C{H_3} - CH = C{\left( {C{H_3}} \right)_2},$$ $$Y = HCHO,$$ $$Z = C{H_3}CHO$$
Answer :
$$X = C{H_3} - CH = CH - C{H_3},$$ \[Y=C{{H}_{3}}\overset{\begin{smallmatrix}
C{{H}_{3}} \\
|\,\,\,\,\,
\end{smallmatrix}}{\mathop{-CH-}}\,CHO,\] $$Z = HCHO$$
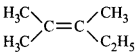 will not show geometrical isomerism as it has two \[-C{{H}_{3}}\] groups on the same carbon atom of the double bond.
will not show geometrical isomerism as it has two \[-C{{H}_{3}}\] groups on the same carbon atom of the double bond.