91. Which of the following statements is not true?
A
$${\left( {C{H_3}} \right)_2}NH$$ is a stronger base than $${\left( {C{H_3}} \right)_3}N.$$
B
Secondary amines show carbylamine reaction.
C
Nitrogen gas is evolved when ethylamine is treated with nitrous acid.
D
Primary amines can show carbylamine reaction.
Answer :
Secondary amines show carbylamine reaction.
92. The correct order of boiling points of the following isomeric amines is $${C_4}{H_9}N{H_2},{\left( {{C_2}{H_5}} \right)_2}NH,$$ $${C_2}{H_5}N{\left( {C{H_3}} \right)_2}$$
A
$${C_2}{H_5}N{\left( {C{H_3}} \right)_2} > {\left( {{C_2}{H_5}} \right)_2}NH$$ $$ > {C_4}{H_9}N{H_2}$$
B
$${\left( {{C_2}{H_5}} \right)_2}NH > {C_2}{H_5}N{\left( {C{H_3}} \right)_2}$$ $$ > {C_4}{H_9}N{H_2}$$
C
$${C_4}{H_9}N{H_2} > {\left( {{C_2}{H_5}} \right)_2}NH > $$ $${C_2}{H_5}N{\left( {C{H_3}} \right)_2}$$
D
$${\left( {{C_2}{H_5}} \right)_2}NH > {C_4}{H_9}N{H_2} > $$ $${C_2}{H_5}N{\left( {C{H_3}} \right)_2}$$
Answer :
$${C_4}{H_9}N{H_2} > {\left( {{C_2}{H_5}} \right)_2}NH > $$ $${C_2}{H_5}N{\left( {C{H_3}} \right)_2}$$
93. Mark the correct statement.
A
Methyl amine is slightly acidic
B
Methyl amine is less basic than ammonia
C
Methyl amine is a stronger base than \[N{{H}_{3}}\]
D
Methyl amine forms salts with alkalies
Answer :
Methyl amine is a stronger base than \[N{{H}_{3}}\]
94.
Identify $$Z$$ in the following sequence of reactions –
\[C{{H}_{3}}COON{{H}_{4}}\xrightarrow{\Delta }X\xrightarrow{{{P}_{2}}{{O}_{5}}}Y\xrightarrow{{{H}_{2}}O/H}Z\]
A
\[C{{H}_{3}}-C{{H}_{2}}-CO-N{{H}_{2}}\]
B
\[C{{H}_{3}}-CN\]
C
\[{{\left( C{{H}_{3}}CO \right)}_{2}}O\]
D
\[C{{H}_{3}}-COOH\]
Answer :
\[C{{H}_{3}}-COOH\]
95. Which of the following is not a property of diazonium salts?
A
Diazonium salts are colourless crystalline solids.
B
Being ionic in nature they are soluble in water.
C
Most of these salts explode when dried.
D
The aqueous solutions of these salts are poor conductors of electricity.
Answer :
The aqueous solutions of these salts are poor conductors of electricity.
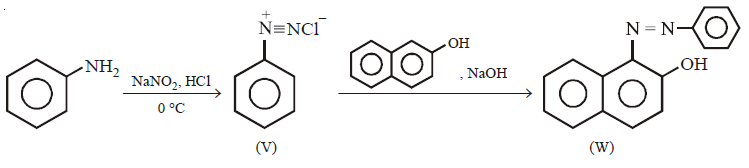
96.
In the following reactions, the major product $$W$$ is
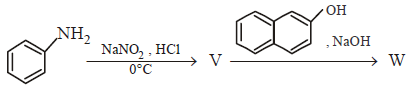
A
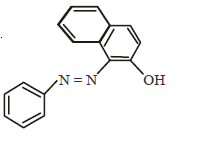

B
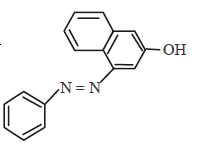

C
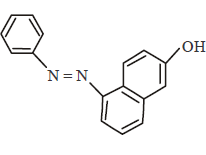

D
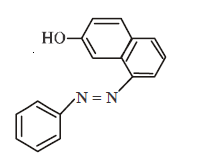

Answer :


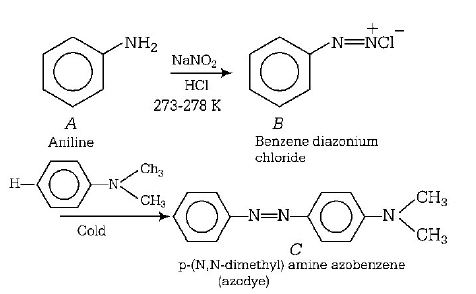
97.
In a reaction of aniline a coloured products $$C$$ was obtained.
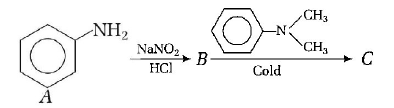
The structure of $$C$$ would be
A
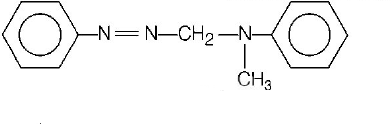

B
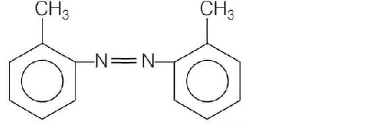

C
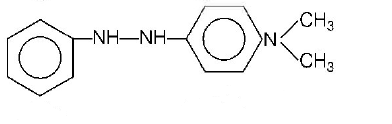

D
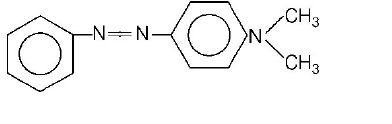

Answer :


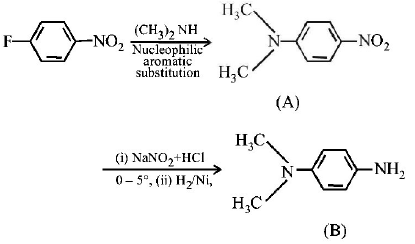
98.
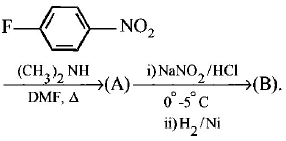 $$B$$ is :
$$B$$ is :
A
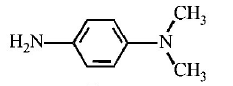

B
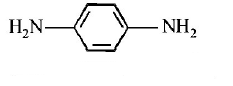

C
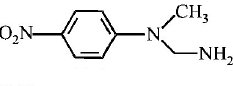

D
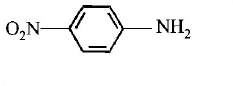

Answer :


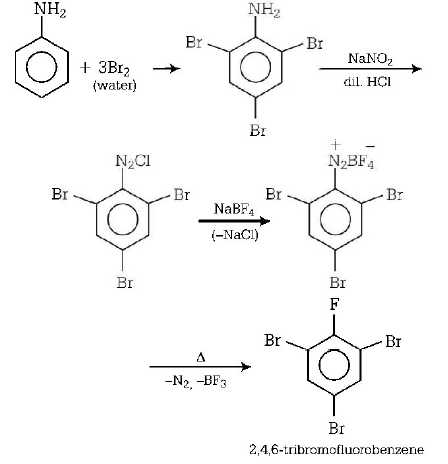
99. Aniline is reacted with bromine water and the resulting product is treated with an aqueous solution of sodium nitrite in presence of dilute hydrochloric acid. The compound so formed is converted into a tetrafluoroborate which is subsequently heated. The final product is
A
1, 3, 5 - tribromobenzene
B
$$p$$ - bromofluorobenzene
C
$$p$$ - bromoaniline
D
2, 4, 6 - tribromofluorobenzene
Answer :
2, 4, 6 - tribromofluorobenzene
100. On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is :
A
an alkanol
B
an alkanediol
C
an alkyl cyanide
D
an alkyl isocyanide
Answer :
an alkyl isocyanide