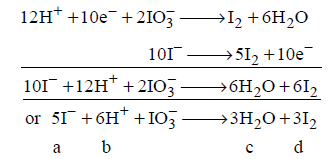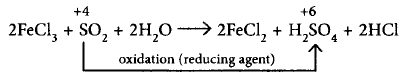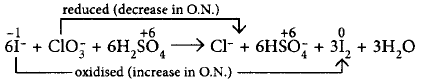151. Which of the following statements is not true?
A
In a chemical reaction, oxidation is always accompanied by reduction.
B
When a negative ion changes to neutral species, the process is oxidation.
C
Oxidising agent has a tendency to lose electrons.
D
Conversion of Conversion of $$MnO_4^{2 - }$$ to $$MnO_4^ - $$ is oxidation.
Answer :
Oxidising agent has a tendency to lose electrons.
152. In the conversion of $$B{r_2}$$ to $$BrO_3^ - ,$$ the oxidation state of $$Br$$ changes from
A
+1 to +5
B
0 to -3
C
+2 to +5
D
0 to +5
Answer :
0 to +5
153. When $$Mn{O_2}$$ salt is fused with a mixture of $$KN{O_3}$$ and solid $$KOH$$ the oxidation number of $$Mn$$ changes from + 4 to
A
+ 4
B
+ 3
C
+ 6
D
+ 7
Answer :
+ 6
154. Which of the following is not a redox reaction?
A
$${P_4} + 3O{H^ - }3{H_2}O \to P{H_3} + 3{H_2}PO_2^ - $$
B
$$Na + {H_2}O \to NaOH + \frac{1}{2}{H_2}$$
C
$$CaC{O_3} \to CaO + C{O_2}$$
D
$${V_2}{O_5} + 5Ca \to 2V + 5CaO$$
Answer :
$$CaC{O_3} \to CaO + C{O_2}$$
155.
In the balanced chemical reaction $$IO_3^ - + a{I^ - } + b{H^ - } \to c{H_2}O + d{I_2}$$
$$a, b, c$$ and $$d,$$ respectively, correspond to
A
5, 6, 3, 3
B
5, 3, 6, 3
C
3, 5, 3, 6
D
5, 6, 5, 5
Answer :
5, 6, 3, 3
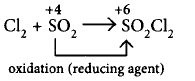
156.
Which of the following reactions shows the reducing property of $$S{O_2}?$$
$$\left( {\text{i}} \right)C{l_2} + S{O_2} \to S{O_2}C{l_2}$$
$$\left( {{\text{ii}}} \right)2{H_2}S + S{O_2} \to 3S + 2{H_2}O$$
$$\left( {{\text{iii}}} \right)2FeC{l_3} + S{O_2} + 2{H_2}O \to $$ $$2FeC{l_2} + {H_2}S{O_4} + 2HCl$$
$$\left( {{\text{iv}}} \right)S{O_2} + {H_2}O \to {H_2}S{O_3}$$
A
(i) only
B
(ii) and (iii)
C
(ii) and (iv)
D
(i), (ii) and (iv)
Answer :
(ii) and (iii)
157. Which of the following elements does not show disproportionation tendency?
A
$$Cl$$
B
$$Br$$
C
$$F$$
D
$$I$$
Answer :
$$F$$
158. Which of the following represents a redox reaction?
A
$$NaOH + HCl \to NaCl + {H_2}O$$
B
$$BaC{l_2} + {H_2}S{O_4} \to BaS{O_4} + 2HCl$$
C
$$CuS{O_4} + 2{H_2}O \to Cu{\left( {OH} \right)_2} + {H_2}S{O_4}$$
D
$$Zn + 2HCl \to ZnC{l_2} + {H_2}$$
Answer :
$$Zn + 2HCl \to ZnC{l_2} + {H_2}$$
159.
For the reaction : $${I^ - } + ClO_3^ - + {H_2}S{O_4} \to $$ $$C{l^ - } + HSO_4^ - + {I_2}$$
The incorrect statement in the balanced equation is
A
stoichiometric coefficient of $$HSO_4^ - $$ is 6
B
iodide is oxidised
C
sulphur is reduced
D
$${H_2}O$$ is one of the products
Answer :
sulphur is reduced
160. Oxidation number of sulphur in peroxomono-sulphuric acid $$\left( {{H_2}S{O_5}} \right)$$ is
A
+ 4
B
+ 2
C
+ 6
D
- 2
Answer :
+ 6