21. The oxidation number of an element in a compound is evaluated on the basis of certain rules. Which of the following rules is not correct in this respect?
A
The oxidation number of hydrogen is always + 1.
B
The algebraic sum of all the oxidation numbers in a compound is zero.
C
An element in the free or the uncombined state bears oxidation number zero.
D
In all its compounds, the oxidation number of fluorine is - 1.
Answer :
The oxidation number of hydrogen is always + 1.
22. Fluorine is best oxidising agent because
A
it is most electronegative.
B
it has highest reduction potential.
C
it has highest oxidation potential.
D
it has smallest size.
Answer :
it has highest reduction potential.
23. Which of the following reactions takes place at anode?
A
Reduction
B
Oxidation
C
Decomposition
D
Dissolution
Answer :
Oxidation
24. In which of the following pairs, there is greatest difference in the oxidation number of the underlined elements ?
A
$$\underline N {O_2}\,{\text{and}}\,\underline N {\,_2}{O_4}$$
B
$${\underline P _{\,2}}{O_5}\,{\text{and}}\,{\underline P _{\,4}}{O_{10}}$$
C
$${\underline N _2}O\,{\text{and}}\,\underline N O$$
D
$$\underline S {O_2}\,{\text{and}}\,\underline S {O_3}$$
Answer :
$$\underline S {O_2}\,{\text{and}}\,\underline S {O_3}$$
25.
How many electrons are involved in the following redox reaction?
$$C{r_2}O_7^{2 - } + F{e^{2 + }} + {C_2}O_4^{2 - } \to $$ $$C{r^{3 + }} + F{e^{3 + }} + C{O_2}\left( {{\text{Unbalanced}}} \right)$$
A
3
B
4
C
6
D
5
Answer :
3
26.
Given below is the set up for Daniell cell. Label $$p, q, r, s, t$$ in the given figure.
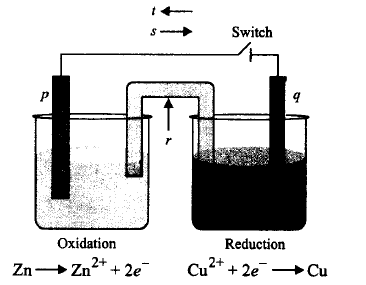
| $$p$$ | $$q$$ | $$r$$ | $$s$$ | $$t$$ | |
| (a) | Anode | Cathode | Salt bridge | Electron flow | Current flow |
| (b) | Cathode | Anode | Salt bridge | Current flow | Electron flow |
| (c) | Anode | Cathode | Salt bridge | Current flow | Electron flow |
| (d) | Cathode | Anode | Salt bridge | Ions flow | Electron flow |
A
(a)
B
(b)
C
(c)
D
(d)
Answer :
(a)
27.
Match the column I with column II and mark the appropriate choice.
| Column I (compound) | Column II (Oxidation state of $$Fe$$) | ||
|---|---|---|---|
| a. | $${K_3}\left[ {Fe{{\left( {OH} \right)}_6}} \right]$$ | 1. | $$ + \frac{8}{3}$$ |
| b. | $${K_2}\left[ {Fe{O_4}} \right]$$ | 2. | $$ + 2$$ |
| c. | $$FeS{O_4} \cdot {\left( {N{H_4}} \right)_2}S{O_4} \cdot 6{H_2}O$$ | 3. | $$ + 3$$ |
| d. | $$F{e_3}{O_4}$$ | 4. | $$ + 6$$ |
A
a - 3, b - 1, c - 2, d - 4
B
a - 3, b - 4, c - 2, d - 1
C
a - 1, b - 3, c - 2, d - 4
D
a - 4, b - 2, c - 1, d - 3
Answer :
a - 3, b - 4, c - 2, d - 1
28.
In the following balanced reaction,
$$X\,MnO_4^ - + Y\,{C_2}O_4^{2 - } + Z\,{H^ + } \rightleftharpoons $$ $$X\,M{n^2} + 2Y\,C{O_2} + \frac{Z}{2}{H_2}O$$
values of $$X, Y$$ and $$Z$$ respectively are
A
2, 5, 16
B
8, 2, 5
C
5, 2, 16
D
5, 8, 4
Answer :
2, 5, 16
29.
Arrange the following in increasing order of oxidation state of $$Ni.$$
$${K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right],{K_2}\left[ {Ni{F_6}} \right],Ni{\left( {CO} \right)_4}$$
A
$$Ni{\left( {CO} \right)_4},{K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right],$$ $${K_2}\left[ {Ni{F_6}} \right]$$
B
$${K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right],Ni{\left( {CO} \right)_4},$$ $${K_2}\left[ {Ni{F_6}} \right]$$
C
$$Ni{\left( {CO} \right)_4},{K_2}\left[ {Ni{F_6}} \right],$$ $${K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right]$$
D
$${K_2}\left[ {Ni{F_6}} \right],{K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right],$$ $$Ni{\left( {CO} \right)_4}$$
Answer :
$$Ni{\left( {CO} \right)_4},{K_2}\left[ {Ni{{\left( {CN} \right)}_4}} \right],$$ $${K_2}\left[ {Ni{F_6}} \right]$$
30. Which of the following is not a correct statement about electrochemical series of reduction potentials ?
A
The standard electrode potential of hydrogen is 0.00 volts.
B
Active non-metals have positive reduction potentials.
C
Active metals have negative reduction potentials.
D
Metals which have positive reduction potentials are good reducing agent.
Answer :
Metals which have positive reduction potentials are good reducing agent.