31.
Arrange the following metals in which they displace each other from the solutions of their salts in decreasing order.
$$Al,Cu,Fe,Mg$$ and $$Zn$$
$$\left[ {E_{\frac{{A{l^{3 + }}}}{{Al}}}^ \circ = - 1.66\,V,E_{\frac{{C{u^{2 + }}}}{{Cu}}}^ \circ = + 0.34\,V,E_{\frac{{F{e^{2 + }}}}{{Fe}}}^ \circ = - 0.44\,V,E_{\frac{{M{g^{2 + }}}}{{Mg}}}^ \circ = - 2.36\,V\,\,{\text{and}}\,\,E_{\frac{{Z{n^{2 + }}}}{{Zn}}}^ \circ = - 0.76\,V} \right]$$
A
$$Cu,Fe,Zn,Al,Mg$$
B
$$Fe,Zn,Cu,Al,Mg$$
C
$$Mg,Cu,Fe,Zn,Al$$
D
$$Mg,Al,Zn,Fe,Cu$$
Answer :
$$Mg,Al,Zn,Fe,Cu$$
32. Which of the following is not a disproportionation reaction?
A
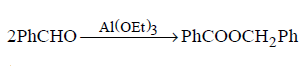
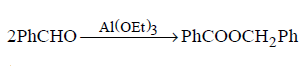
B
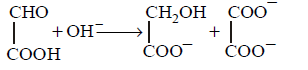
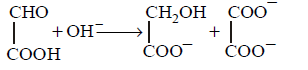
C
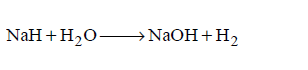
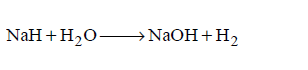
D
Answer :
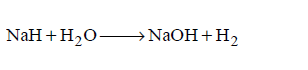
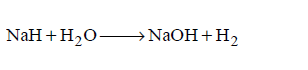
33. Which of the following is the strongest reducing agent in aqueous medium?
A
$$Mg$$
B
$$Na$$
C
$$Li$$
D
$$Ca$$
Answer :
$$Li$$
34. Which of the following colour changes shown during redox titrations is not correct?
A
$$C{r_2}O_7^{2 - }$$ oxidises the indicator diphenylamine to produce blue colour showing end point.
B
Iodine formed by oxidation of $${I^ - }$$ ions gives blue colour with starch showing end point.
C
$$KMn{O_4}$$ in the form of $$MnO_4^ - $$ ions gives pink colour showing end point.
D
Thiosulphate ions $$\left( {{S_2}O_3^{2 - }} \right)$$ give blue colour showing end point.
Answer :
Thiosulphate ions $$\left( {{S_2}O_3^{2 - }} \right)$$ give blue colour showing end point.
35. Which of the following is not an intermolecular redox reaction?
A
$$MgC{O_3} \to MgO + C{O_2}$$
B
$${O_2} + 2{H_2} \to 2{H_2}O$$
C
$$K + {H_2}O \to KOH + \left( {\frac{1}{2}} \right){H_2}$$
D
$$MnB{r_3} \to MnB{r_2} + \left( {\frac{1}{2}} \right)B{r_2}$$
Answer :
$$MgC{O_3} \to MgO + C{O_2}$$
36. In which of the following compounds oxidation state of chlorine has two different values?
A
$$CaC{l_2}$$
B
$$NaCl$$
C
$$CaOC{l_2}$$
D
$$CC{l_4}$$
Answer :
$$CaOC{l_2}$$
37. The oxidation number of phosphorus in $$Ba{\left( {{H_2}P{O_2}} \right)_2}$$ is
A
$$+3$$
B
$$+2$$
C
$$+1$$
D
$$-1$$
Answer :
$$+1$$
38.
Which of the following will act as cathode when connected to standard hydrogen electrode which has $${E^ \circ }$$ value given as zero?
$$\eqalign{
& \left( {\text{i}} \right)\frac{{Z{n^{2 + }}}}{{Zn}},{E^ \circ } = - 0.76\,V \cr
& \left( {{\text{ii}}} \right)\frac{{C{u^{2 + }}}}{{Cu}},{E^ \circ } = + 0.34\,V \cr
& \left( {{\text{iii}}} \right)\frac{{A{l^{3 + }}}}{{Al}},{E^ \circ } = - 1.66\,V \cr
& \left( {{\text{iv}}} \right)\frac{{H{g^{2 + }}}}{{Hg}},{E^ \circ } = + 0.885\,V \cr} $$
A
(i) and (ii)
B
(ii) and (iv)
C
(i) and (iii)
D
(i), (ii), (iii) and (iv)
Answer :
(ii) and (iv)
39. The correct decreasing order of oxidation number of oxygen in compounds $$Ba{O_2},{O_3},K{O_2}$$ and $$O{F_2}$$ is :
A
$$Ba{O_2} > K{O_2} > {O_3} > O{F_2}$$
B
$$O{F_2} > {O_3} > K{O_2} > Ba{O_2}$$
C
$$K{O_2} > O{F_2} > {O_3} > Ba{O_2}$$
D
$$Ba{O_2} > {O_3} > O{F_2} > K{O_2}$$
Answer :
$$O{F_2} > {O_3} > K{O_2} > Ba{O_2}$$
40.
What is the correct representation of reaction occurring when $$HCl$$ is heated with $$Mn{O_2}?$$
A
$$MnO_4^ - + 5C{l^ - } + 8{H^ + } \to $$ $$M{n^{2 + }} + 5C{l^ - } + 5{H_2}O$$
B
$$Mn{O_2} + 2C{l^ - } + 4{H^ + } \to $$ $$M{n^{2 + }} + C{l_2} + 2{H_2}O$$
C
$$2Mn{O_2} + 4C{l^ - } + 8{H^ + } \to $$ $$2M{n^{2 + }} + 2C{l_2} + 4{H_2}O$$
D
$$Mn{O_2} + 4HCl \to MnC{l_4} + C{l_2} + {H_2}O$$
Answer :
$$Mn{O_2} + 2C{l^ - } + 4{H^ + } \to $$ $$M{n^{2 + }} + C{l_2} + 2{H_2}O$$