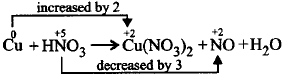41.
The oxidation number of $$Cr$$ in $$Cr{O_5}$$ which has the following structure is 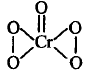
A
+4
B
+5
C
+6
D
+3
Answer :
+6
42.
The stoichiometric constants for the reaction $$pCu + qHN{O_3} \to $$ $$rCu{\left( {N{O_3}} \right)_2} + sNO + t{H_2}O$$
$$p,q,r,s$$ and $$t$$ respectively are
A
3, 3, 3, 2, 3
B
3, 2, 3, 2, 4
C
3, 8, 3, 2, 4
D
2, 3, 3, 3, 2
Answer :
3, 8, 3, 2, 4
43. The oxidation state of $$Fe$$ in $${K_4}\left[ {Fe{{\left( {CN} \right)}_6}} \right]$$ is
A
+ 2
B
+ 3
C
+ 4
D
+ 6
Answer :
+ 2
44.
Given $$E_{\frac{{A{g^ + }}}{{Ag}}}^ \circ = + 0.80\,V;E_{\frac{{C{u^{2 + }}}}{{Cu}}}^ \circ = + 0.34\,V;$$ $$E_{\frac{{F{e^{3 + }}}}{{F{e^{2 + }}}}}^ \circ = + 0.76\,V;E_{\frac{{C{e^{4 + }}}}{{C{e^{3 + }}}}}^ \circ = + 1.60\,V$$
Which of the following statements is not correct?
A
$$F{e^{3 + }}$$ does not oxidise $$C{e^{3 + }}.$$
B
$$Cu$$ reduces $$A{g^ + }$$ to $$Ag.$$
C
$$Ag$$ will reduce $$C{u^{2 + }}$$ to $$Cu.$$
D
$$F{e^{3 + }}$$ reduces $$C{u^{2 + }}$$ to $$Cu.$$
Answer :
$$Ag$$ will reduce $$C{u^{2 + }}$$ to $$Cu.$$
45. Which halide is not oxidised by $$Mn{O_2}?$$
A
$$F$$
B
$$Cl$$
C
$$Br$$
D
$$I$$
Answer :
$$F$$
46. The pair of compounds in which the metals are in their highest oxidation state is
A
$$Mn{O_2},FeC{l_3}$$
B
$$MnO_4^ - ,Cr{O_2}C{l_2}$$
C
$$MnC{l_2},CrC{l_3}$$
D
$${\left[ {NiC{l_4}} \right]^{2 - }},{\left[ {CoC{l_4}} \right]^ - }$$
Answer :
$$MnO_4^ - ,Cr{O_2}C{l_2}$$
47. In a balance equation $${H_2}S{O_4} + xHI \to {H_2}S + y{I_2} + z{H_2}O,$$ the values of $$x,y,z$$ are
A
$$x = 3,y = 5,z = 2$$
B
$$x = 4,y = 8,z = 5$$
C
$$x = 8,y = 4,z = 4$$
D
$$x = 5,y = 3,z = 4$$
Answer :
$$x = 8,y = 4,z = 4$$
48. In the reaction : $${I_2} + 2{S_2}O_3^{2 - } \to 2{I^ - } + {S_4}O_6^{2 - }$$
A
$${I_2}$$ is reducing agent.
B
$${I_2}$$ is oxidising agent and $${S_2}O_3^{2 - }$$ is reducing agent.
C
$${S_2}O_3^{2 - }$$ is oxidising agent.
D
$${I_2}$$ is reducing agent and $${S_2}O_3^{2 - }$$ is oxidising agent.
Answer :
$${I_2}$$ is oxidising agent and $${S_2}O_3^{2 - }$$ is reducing agent.
49.
Using the following Latimer diagram for bromine, \[pH=0;BrO_{4}^{-}\xrightarrow{1.82\,V}BrO_{3}^{-}\] \[\xrightarrow{1.50\,V}HBrO\xrightarrow{1.595\,V}B{{r}_{2}}\] \[\xrightarrow{1.06552\,V}B{{r}^{-}}\]
the species undergoing disproportionation is
A
$$BrO_4^ - $$
B
$$BrO_3^ - $$
C
$$HBrO$$
D
$$B{r_2}$$
Answer :
$$HBrO$$
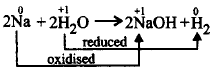
50. When a piece of sodium metal is dropped in water, hydrogen gas evolved because
A
sodium is reduced and acts as an oxidising agent
B
water is oxidised and acts as a reducing agent
C
sodium loses electrons and is oxidised while water is reduced
D
water loses electrons and is oxidised to hydrogen
Answer :
sodium loses electrons and is oxidised while water is reduced